Pseudomonas aeruginosa is a non-enteric, oxidase positive, Gram-negative, motile; obligate aerobic straight or curved rod that is found in the genus Pseudomonas and family Pseudomonadaceae. They are recurrently present in small numbers as members of the normal flora in the small intestine and skin of humans especially in hospitalized patients. P. aeruginosa is the most important member of the genus Pseudomonas due to its ability to cause infections in man and animals. They are strict aerobes and thus derive their energy through the oxidation of sugars rather than via sugar fermentation as is the case for most members of the Enterobacteriaceae family.
P. aeruginosa like other enteropathogenic E. coli strainshas pathogenicity islands in its genome, and this feature increases its virulence during an infection. Other non-pathogenic Pseudomonas species are used in bioremediation activities and some also degrade organic matters into inorganic substances through the process of mineralization. P. aeruginosa is mostly implicated in cystic fibrosis infection, and also in wound infections, bacteraemia, soft tissue infections, dermatitis, burns, otitis externa (ear infections) and cellulitis. It grows on normal culture media such as blood agar, MacConkey agar and chocolate agar at 35-37oC and they are non-lactose fermenters.
P. aeruginosa is a pigment-producing free-living environmental opportunistic pathogen that is notorious in causing hospital-acquired infections even though they are seen as contaminants and colonizers of moist surroundings. Hospitalized patients with weakened immunity are mostly affected and the pathogen is usually resistant to antimicrobial agents due to its inherent and acquired traits to drugs. P. aeruginosa is uncommonly isolated from non-hospitalized patients, but hospitalization increases the frequency and chances of infection with the pathogen.
P. aeruginosa produces two types of soluble pigments known as pyocyanin (bluish pigment) and fluorescein or pyoverdin (fluorescent-greenish pigment). Pyocyanin pigment is mostly produced in wound infections caused by pyocyanin-producing P. aeruginosa whilepyoverdin(which plays a role in the iron metabolism of the bacterium) is produced in culture media of low-iron content (e.g. blood agar – in which P. aeruginosa forms a blackish metallic sheen). The black pigment produced by some strains of Pseudomonas species on agar is known as pyorubin or pyomelanin.
P. aeruginosa (the leading cause of nosocomial infections) is also implicated in soft tissue infections, urinary tract infections and pneumonia. They are ubiquitous and widely distributed in nature and can be found in the soil, plants, animals and water. Swimming pools and whirlpools with high colony counts of P. aeruginosa are the main source through which people acquire hot-tub folliculitis. In hospital environment, they are known to colonize moist environments such as washing sinks, pipes, washing and cleaning buckets, and drains.
PATHOGENESISOF PSEUDOMONAS AERUGINOSA INFECTION
P. aeruginosa gains entry into the human body via injury or damage on the skin and the mucous membranes of internal body tissues. A significant opening or fracture in the first-line of defenses of the human body (e.g. the skin) is the main route via which P. aeruginosa enters the body. It can also enter through contaminated invasive medical devices or products such as intra-tracheal devices, distilled water, anesthesia apparatus, and urinary catheters. P. aeruginosa can also form biofilms on plastic implants. Generally, an alteration of the normal defense mechanisms of the human host initiates infection or disease by opportunistic microbes such as P. aeruginosa.
Upon entry, P. aeruginosa attaches to the mucous membranes and/or epithelial cells of internal body tissues using its pili and colonizes the region to cause systemic infections. Its pathogenesis though highly complex and multifaceted is usually based on the ability of the pathogen to produce several virulence factors such as toxins, adhesions, capsules, pigment production (i.e. pyocyanin), lipopolysaccharides (LPS) and enzymes which enhances its disease process and underscores the reason why P. aeruginosa is implicated in a wide variety of human diseases. These virulence factors also break down physical barriers and damage host cells.
Tissue necrosis, sepsis, and septic shock are some of the symptoms associated with P. aeruginosa infection. Other clinical findings in which P. aeruginosa is implicated include otitis externa (in swimmers ear), osteomyelitis, corneal/eye infections, endocarditis (in intravenous drug users), respiratory infections, folliculitis of the skin (after soaking body in poorly decontaminated hot tubs heavily contaminated with P. aeruginosa) and ecthyma gangrenosum (lesions formed due to haemorrhagic necrosis of the skin caused by P. aeruginosa sepsis).
Though the pathogen can cause infection in any part of the human body; wound infections (caused by burns), pneumonia (in cystic fibrosis patients) and urinary tract infections are usually the most predominating diseases in which P. aeruginosa is implicated in humans. P. aeruginosa is a major threat to cystic fibrosis (CF) patients and the most persistent pathogen that complicates the clinical course of CF disease because the pathogen produces pigments (pyocyanin) that damages the cilliary and mucosal actions of the respiratory tract epithelial cells, thus eliminating or impairing its normal clearance mechanisms and retarding desquamation in the region.
P. aeruginosa infection in CF patients form biofilms that contains microcolonies of bacterial cells that protects the pathogen from antimicrobial onslaught and attack by the host immune system. Immunocompromised individuals (e.g. cancer and HIV patients), malnourished persons and people already in poor health and with weakened immune system are mostly the ones affected by P. aeruginosa. P. aeruginosa is mostly found on the moist areas of the human skin and they also colonize the upper respiratory tract of hospitalized patients.
LABORATORY DIAGNOSIS OF PSEUDOMONAS AERUGINOSA INFECTION
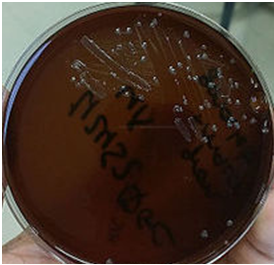
The laboratory diagnosis of P. aeruginosa infection depends largely on microscopy, isolation and identification of the pathogen from clinically important specimens usually collected based on the type of infection at hand. P. aeruginosa grow effectively on MacConkey agar producing pale coloured colonies (Figure 1), CLED medium (producing green colonies), and on blood agar (producing large, flat spreading colonies that can be haemolytic, mucoid or pigment-producing).
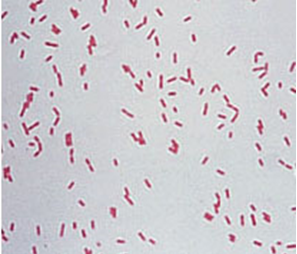
The specimens collected for the detection and isolation of P. aeruginosa include pus, wound specimens, urine, CSF, skin lesions, and blood. The Gram reaction of Pseudomonas aeruginosa is negative (Figure 2). Biochemically, P. aeruginosa isolates are oxidase positive and they do not ferment lactose. P. aeruginosa produces a characteristic pungent (fruity) odour in cultures due to the production of 2-aminoacetophenone by the pathogen during growth.
P. aeruginosa grows at temperature range of 37-42oC; and its ability to grow at 42oC helps to differentiate the pathogen from other Pseudomonas species. Fluorescent test conducted under UV light can be used in the preliminary identification of the pathogen from cultures since P. aeruginosa produces fluorescein and thus fluoresces under the light.
IMMUNITY TO PSEUDOMONAS AERUGINOSA INFECTION
Protection against some P. aeruginosa infections (with the exception of cystic fibrosis) in individuals with strong immunity is usually based on opsonization and phagocytosis. Antibody action and other components of the immune system especially the humoural immunity are unleashed against the pathogen in vivo once an infection has been established.
TREATMENT OF PSEUDOMONAS AERUGINOSA INFECTION
Most of the commonly used antibiotics including penicillin, chloramphenicol, ampicillin, tetracycline, sulphonamides, some aminoglycosides, and first and second-generation cephalosporins due to the nature of its efflux-porin pumps in restricting or actively pumping out antibiotics before they reach the periplasmic space of the pathogen. Thus, the treatment of P. aeruginosa infection should be based on the availability of a proper susceptibility test results to select the best drugs. Therapy should not be also based on a single antibiotic regimen due to the multidrug resistant nature of the pathogen. Drugs proven to be active against P. aeruginosa include third-generation cephalosporins, some aminoglycosides, fluoroquinolones, and the carbapenems. Effective oral antibiotics for cystic fibrosis patients when P. aeruginosa is implicated are scarce as most agents used are given parenterally.
PREVENTION AND CONTROL OF PSEUDOMONAS AERUGINOSA INFECTION
P. aeruginosa is an opportunistic pathogen that is notorious in causing hospital-associated diseases. Since the pathogen is a common inhabitant of moist environments including water, soil, vegetations, hospital surfaces, toilets, sinks, and medical devices/equipments and reagents, attention should be given to these areas in terms of ensuring their proper disinfection. Infected patients and hospital staff carrying P. aeruginosa are primary sources of infection as infection can occur from patient to patient and via the hands of infected hospital workers. Direct contact with contaminated reservoirs of the pathogen and consumption of contaminated food and water can also lead to an infection with P. aeruginosa. Thus, proper personal hygiene and infection control practices should be imbibed by hospital staff, patients and visitors to hospital settings.
OTHER SPECIES OF PSEUDOMONAS
- P. putida: P. putida is found in most soil and water habitats where there is oxygen; and it has application in bioremediation activities. Most of its strains are non-pathogenic.
- P.aureofaciens: P.aureofaciens is a soil-borne root-colonizing bacterium that produces phenazine, an antimicrobial agent. Phenazine production is the primary mechanism responsible for the competitive fitness of P. aureofaciens in the rhizosphere of the soil.
- P. stutzeri: P. stutzeri is a denitrifying bacterium that fixes dinitrogen in the soil; and it is used for bioremediation activities. It is an opportunistic organism commonly isolated from clinical settings and humans.
- P. mallei: P. mallei is now known as Burkholderia mallei; and Pseudomonas (Burkholderia) mallei is causative agents of melioidosis and glanders that affects animals such as horses, mules and donkeys. Horses are the natural host of B. mallei; and human infections occur from them.
- P. pseudomallei: P. pseudomallei is also known as Burkholderia pseudomallei even though B. pseudomallei was previously classified as part of the Pseudomonas genus. The organism is now known as B. pseudomallei. P. pseudomallei is a soil-dwelling bacterium endemic in tropical and subtropical regions worldwide; and it infects humans and animals and causes the disease melioidosis. The organism also infects plants.
- P. denitrificans: P. denitrificans is distributed in the soil and it synthesizes vitamin B12. It has the ability to perform denitrification in the soil.
- P. syringae: P. syringae is a plant pathogen known for its capacity to grow epiphytically on diverse plants and woods. It can also be found in non-agricultural habitats, particularly those associated with water; and it attacks plants or woods affected by frost/ice.
- P. fluorescens: P. fluorescens is a nonpathogenic saprophyte that colonizes the soil, water and plant surface environments; and the organism is not generally considered a bacterial pathogen in humans, though it occurs as indigenous microbiota of various body sites in low levels. Medically, cultures of P. fluorescens produce mupirocin, an antibiotic that is used to treat skin and eye infections. The organism produces compounds antagonistic to other soil microbes; and it can be used as a biocontrol agent since is antagonistic compounds protect the roots of some plant species against parasitic fungi such as Fusarium species.
References
Brooks G.F., Butel J.S and Morse S.A (2004). Medical Microbiology, 23rd edition. McGraw Hill Publishers. USA. Pp. 248-260.
Madigan M.T., Martinko J.M., Dunlap P.V and Clark D.P (2009). Brock Biology of microorganisms. 12th edition. Pearson Benjamin Cummings Publishers. USA. Pp.795-796.
Prescott L.M., Harley J.P and Klein D.A (2005). Microbiology. 6th ed. McGraw Hill Publishers, USA. Pp. 296-299.
Ryan K, Ray C.G, Ahmed N, Drew W.L and Plorde J (2010). Sherris Medical Microbiology. Fifth edition. McGraw-Hill Publishers, USA.
Singleton P and Sainsbury D (1995). Dictionary of microbiology and molecular biology, 3rd ed. New York: John Wiley and Sons.
Talaro, Kathleen P (2005). Foundations in Microbiology. 5th edition. McGraw-Hill Companies Inc., New York, USA.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.