Antibiotics can also be classified into different categories depending on their mode of action and/or spectrum of activity.
BETA-LACTAM ANTIBIOTICS
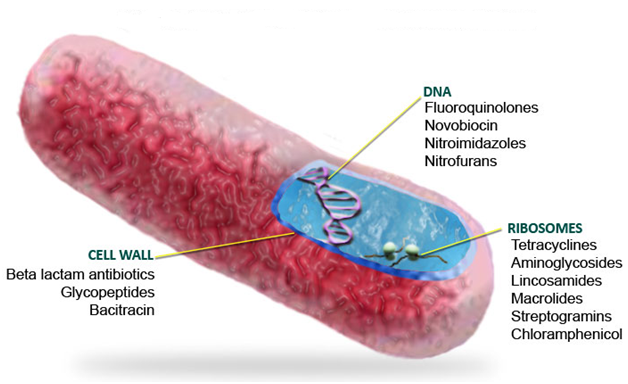
Beta-lactam antibiotics are group of naturally-synthesized antibiotics that inhibit the synthesis of cell wall in bacteria. Some later beta-lactam drugs such as methicillin, ampicillin and amoxicillin amongst others are synthesized by chemical processes. Beta-lactam drugs contain a 4-membered beta lactam ring; and this beta-lactam ring is usually the main target of some antibiotic degrading enzymes such as the beta-lactamases. The 4-membered beta-lactam ring is the main component of the structure of all beta-lactam antibiotics. Antibiotics in this category bind to the penicillin-binding-proteins (PBPs) of the bacterial cell, and this binding interferes with the transpeptidases enzymes involved in transpeptidation reaction- which is vital for the cross-linking of N-acetyl muramic acid (NAM) and N-acetyl glucosamine (NAG) molecules required for the formation of peptidoglycan (that maintains the integrity of the bacterial cell through the formation of cell wall). Beta-lactam antibiotics are bacteriocidal, and thus they kill bacterial cells by preventing the formation of cell walls.
Prevention of the formation of a cell wall leaves a bacteria cell porous to attack from its environment, and this leads to lysis of the cell due to external pressure from the outside. And this could be as a result of differences in the osmotic pressure of the internal environment of the cell and that of its external environment which allows different molecules to gain entry into the cell’s internal environment. Bacterial cells could swell and rupture or lyse (i.e. die) in this manner. Typical examples of antibiotics that are beta-lactams are penicillins and cephalosporins which are mainly produced naturally by two fungi genera: Penicillium and Cephalosporium. Beta-lactam antibiotics inhibit some basic steps in the synthesis of cell wall in bacterial cell especially as it relates to the peptidoglycan and murein components. They are active against both Gram positive and Gram negative bacteria; and thus beta-lactam drugs are bacteriocidal in action.
Macrolides include erythromycin and azithromycin; and they are naturally-synthesized antibiotics produced by Streptomyces species (e.g. Streptomyces erythreus). They specifically inhibit translation i.e. protein synthesis in bacterial cells. Antibiotics that are macrolides are known to contain large lactone rings that are linked through glycoside bonds with amino sugars; and this feature differentiate them from beta-lactam drugs that contain a 4-membered beta-lactam ring. Macrolides are mostly bacteriostatic agents, but some are cidal in action against some Gram positive bacteria. Lincomycin and clindamycin are other examples of macrolides that also inhibit protein synthesis in bacterial cells. Macrolides generally inhibit bacterial protein synthesis by binding to the 50S ribosomal subunit; and this interferes with the activity of peptidyl transferase which is supposed to stimulate the elongation of the protein molecules in the bacterial cell. Chloramphenicol is another example of antibiotic that target the 50S ribosomal subunit of bacteria.
QUINOLONES
Quinolones include nalidixic acid, oxolinic acid and conoxacin. Quinolones are synthetic antibiotics that have activity against Gram negative bacteria; and they mainly inhibit DNA replication in bacterial cells by targeting the key enzymes (e.g. DNA gyrase enzyme or topoisomerases) responsible for the biosynthesis of DNA molecules. They are bacteriocidal in action. Quinolones are mainly used to treat urinary tract infections (UTIs) in humans.
FLUOROQUINOLONES
Fluoroquinolones are fluorinated derivatives of quinolones and typical examples include ciprofloxacin, ofloxacin and norfloxacin. The fluoroquinolones are mainly derived by the addition of a fluorine molecule on the carbon-6 (C-6) of the quinolone molecule. Fluoroquinolones like the quinolones are bacterial DNA replication inhibitors; and they are active against Gram negative bacteria and some Gram positive bacteria. They are synthetic antibiotics; and like the quinolones, fluoroquinolones or 4-quinolones are bacterial DNA replication inhibitors. They inhibit DNA gyrase enzyme and topoisomerase IV which are both required for bacterial DNA replication. Fluoroquinolones are broad spectrum antibiotics and they are bacteriocidal in action; and can be used to treat a wide variety of bacterial infections including but not limited to UTIs, intestinal infections, and lower respiratory tract infections amongst others.
TETRACYCLINES
The tetracyclines are group of antibiotics that block protein synthesis in bacteria by binding to the 30S ribosomal subunit. They are bacteriostatic in action but have a broad spectrum of activity; and members of antibiotics in this category include doxycycline, tetracycline and minocycline. Tetracyclines are structurally made up of four-fused benzene ring to which molecular substitutions are attached to generate a different type of tetracycline with unique pharmacological activity. The tetracyclines are naturally synthesized antibiotics, and they are mainly synthesized by Streptomyces species. However, tetracyclines can still be produced semi-synthetically today.
AMINOGLYCOSIDES
Aminoglycosides are antibiotics that inhibit protein synthesis or translation in bacteria. They specifically bind to the 30S ribosomal subunit of bacterial ribosome. Structurally, aminoglycosides have a six-membered aminocyclitol ring to which amino sugars are attached through a glycosidic bond. The aminoglycosides are naturally synthesized antibiotics and they are mainly produced from fungi such as Streptomyces species which synthesize streptomycin andthe bacteria genus Micromonospora which synthesize gentamicin. Tobramycin, neomycin, amikacin and kanamycin are other examples of aminoglycosides. Aminoglycosides are bacteriocidal in action and they are broad spectrum antibiotics; but they are mainly used to treat infections caused by Gram negative bacteria especially those in the family Enterobacteriaceae. They easily penetrate the cytoplasmic membrane of aerobic bacteria than that of anaerobic bacteria. To be effective for treating infections caused by anaerobic bacteria (e.g. Streptococcus species), aminoglycosides are combined or synergistically used with cell wall inhibitors (e.g. vancomycin and penicillin) which open the cell wall of anaerobic organisms and allow the drug to enter the cell and unleash its antimicrobial activity.
SULPHONAMIDES (anti-metabolites)
Sulphonamides are structural analogues of Para-Aminobenzoic Acid (PABA) which is required for the synthesis of folic acid in bacteria. Folic acid is required as a cofactor for the synthesis of nucleotides, and they also act as building blocks for bacterial DNA and RNA. However, prokaryotic cells (particularly bacteria) synthesize their own folic acid molecules unlike eukaryotic cells which obtain theirs from their food intake; and this makes pathogenic bacteria to be more susceptible to antibiotics that are anti-metabolites. Anti-metabolites inhibit dihydropteroate synthase (the enzyme that catalyzes the conversion of PABA to folic acid or dihydropteroic acid); thus disrupt folic acid synthesis in the bacterial cell. Antibiotics that are sulphonamides specifically compete for PABA in the bacterial cell; and the incorporation of sulphonamide instead of PABA by the cell, inhibits the synthesis of folic acid. Thus DNA and RNA synthesis in the bacteria will be impaired. Sulphamethoxazole, pyrimethamine, and trimethoprim are examples of antibiotics known as sulphonamides.
References
Ashutosh Kar (2008). Pharmaceutical Microbiology, 1st edition. New Age International Publishers: New Delhi, India.
Axelsen P. H (2002). Essentials of Antimicrobial Pharmacology. Humana Press, Totowa, NJ.
Balfour H. H (1999). Antiviral drugs. N Engl J Med, 340, 1255–1268.
Bean B (1992). Antiviral therapy: current concepts and practices. Clin Microbiol Rev, 5, 146–182.
Beck R.W (2000). A chronology of microbiology in historical context. Washington, D.C.: ASM Press.
Champoux J.J, Neidhardt F.C, Drew W.L and Plorde J.J (2004). Sherris Medical Microbiology: An Introduction to Infectious Diseases. 4th edition. McGraw Hill Companies Inc, USA.
Chemotherapy of microbial diseases. In: Chabner B.A, Brunton L.L, Knollman B.C, eds. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York, McGraw-Hill; 2011.
Chung K.T, Stevens Jr., S.E and Ferris D.H (1995). A chronology of events and pioneers of microbiology. SIM News, 45(1):3–13.
Courvalin P, Leclercq R and Rice L.B (2010). Antibiogram. ESKA Publishing, ASM Press, Canada.
Denyer S.P., Hodges N.A and Gorman S.P (2004). Hugo & Russell’s Pharmaceutical Microbiology. 7th ed. Blackwell Publishing Company, USA. Pp.152-172.
Dictionary of Microbiology and Molecular Biology, 3rd Edition. Paul Singleton and Diana Sainsbury. 2006, John Wiley & Sons Ltd. Canada.
Drusano G.L (2007). Pharmacokinetics and pharmacodynamics of antimicrobials. Clin Infect Dis, 45(suppl):89–95.
Engleberg N.C, DiRita V and Dermody T.S (2007). Schaechter’s Mechanisms of Microbial Disease. 4th ed. Lippincott Williams & Wilkins, Philadelphia, USA.
Finch R.G, Greenwood D, Norrby R and Whitley R (2002). Antibiotic and chemotherapy, 8th edition. Churchill Livingstone, London and Edinburg.
Ghannoum MA, Rice LB (1999). Antifungal agents: Mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin Microbiol Rev, 12:501–517.
Gillespie S.H and Bamford K.B (2012). Medical Microbiology and Infection at a glance. 4th edition. Wiley-Blackwell Publishers, UK.
Gordon Y. J, Romanowski E.G and McDermitt A M (2005). A review of antimicrobial peptides and their therapeutic potential as anti-infective drugs. Current Eye Research, 30(7): 505-515.
Hardman JG, Limbird LE, eds. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 10th ed. New York: McGraw-Hill; 2001.
Joslyn, L. J. (2000). Sterilization by Heat. In S. S. Block (Ed.), Disinfection, Sterilization, and Preservation (5th ed., pp. 695-728). Philadelphia, USA: Lippincott Williams and Wilkins.
Katzung, B. G. (2003). Basic and Clinical Pharmacology (9th ed.). NY, US, Lange.
Kontoyiannis D.P and Lewis R.E (2002). Antifungal drug resistance of pathogenic fungi. Lancet. 359:1135–1144.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.