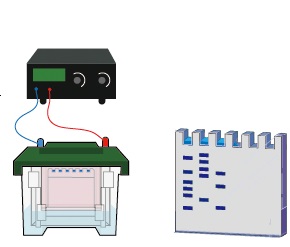
Western blotting technique or protein immunoblot is used to identify specific proteins separated according to their sizes in an electrophoresis experiment. Protein immunoblot unlike other blotting techniques (e.g. southern and northern blotting) utilizes specific antibodies to identify the protein of interest. It is the transfer of proteins from gel to a matrix or nitrocellulose membrane. Western blotting allows molecular biologists to determine the actual molecular weight of a protein molecule and to measure its relative amounts in different samples.
Instead of agarose gel electrophoresis which is usually used for the separation of nucleic acid fragments (particularly those of RNA and DNA), polyacrylamide gel in sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) technique is used to separate the protein fragments prior to the blotting technique. The steps involved in carrying out western blotting or protein immunoblot is highlighted in this unit.
STEPS FOR WESTERN BLOTTING
- Separate the protein sample according to their sizes using SDS-PAGE.
- Transfer the separated proteins from the SDS-PAGE gel to the surface of a nitrocellulose membrane by electroblotting. In electroblotting, electric current is passed through the SDS-PAGE gel, and this transfers the separated proteins onto the nitrocellulose membrane.
- Incubate the separated proteins with a generic protein molecule so that unbound sites of the nitrocellulose paper shall be covered. Addition of generic protein (i.e. protein molecules of known sizes) helps to minimize background signals on the membrane.
- Add specific antibody that is unique to the protein of interest to the protein solution. The antibody (which is usually radioactively labeled) binds to its specific protein molecules on the nitrocellulose membrane. Only the band containing the protein of interest binds to the antibody, thus forming a layer of antibody molecules on the membrane.
- Expose the nitrocellulose membrane to an X-ray film, and photograph it by autoradiography in order to reveal the separated protein bands.
References
Alberts B, Bray D, Lewis J, Raff M, Roberts K and Watson J.D (2002). The molecular Biology of the Cell. Fourth edition. New York, Garland, USA.
Chen I and Dubnau D (2004). DNA uptake during bacterial transformation. Nat. Rev. Microbiol. 2 (3): 241–249.
Cooper G.M and Hausman R.E (2004). The cell: A Molecular Approach. Third edition. ASM Press.
Dale J (2003). Molecular genetics of bacteria. Jeremy W. Dale and Simon Park (4th eds.). John Wiley & Sons Ltd, West Sussex, UK. Pp. 312-313.
Das H.K (2010). Textbook of Biotechnology. Fourth edition. Wiley edition. Wiley India Pvt, Ltd, New Delhi, India.
Lewis R (2004). Human Genetics: Concepts and Applications. Sixth edition. McGraw Hill Publishers, USA.
Lodish H, Berk A, Matsudaira P, Kaiser C.A, Kreiger M, Scott M.P, Zipursky S.L and Darnell J (2004). Molecular Cell Biology. Fifth edition. Scientific American Books, Freeman, New York, USA.
Madigan M.T., Martinko J.M., Dunlap P.V and Clark D.P (2009). Brock Biology of Microorganisms, 12th edition. Pearson Benjamin Cummings Inc, USA.
McPherson M and Moller S (2002). PCR: The Basics. 2nd edition. Taylor and Francis Group. New York, USA.
Sambrook, J., Russell, D.W. (2001). Molecular Cloning: a Laboratory Manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York.
Synder L, Peters J.E, Henkin T.M and Champness W (2013). Molecular Genetics of Bacteria. Fourth edition. American Society of Microbiology Press, USA.
Tamarin Robert H (2002). Principles of Genetics. Seventh edition. Tata McGraw-Hill Publishing Co Ltd, Delhi.
Discover more from #1 Microbiology Resource Hub
Subscribe to get the latest posts to your email.