Nitrate reduction test is used to identify pathogenic bacteria that have the ability to convert nitrate (NO3–) to nitrite (NO2–). Nitrate reduction test is used for the differentiation of members of Enterobacteriaceae on the basis of their ability to produce nitrate reductase enzyme – that hydrolyze nitrate (NO3–) to nitrite (NO2–), which may then again be degraded to various nitrogen products like nitrogen oxide, nitrous oxide and ammonia (NH3) depending on the enzyme system of the test organism and the atmosphere in which it is growing. Nitrate broth is used to determine the ability of an organism to reduce nitrate (NO3) to nitrite (NO2) using the enzyme nitrate reductase.
It also tests the ability of organisms to perform nitrification on nitrate and nitrite to produce molecular nitrogen. The nitrate reductase test is a test to differentiate between bacteria based on their ability or inability to reduce nitrate (NO3−) to nitrite (NO2−) using anaerobic respiration. Certain Gram negative and Gram positive bacteria produce the enzyme, nitrate reductase (nitratase) which reduces nitrate to other nitrogenous compounds including NO2–, and this serves as basis for their identification and differentiation from other bacteria that cannot reduce nitrate in the laboratory.
Since the test is performed with Durham tube(s), nitrate reduction test can also be used to detect the production of gases (e.g. nitrogen, N2) by non-fermenters which convert nitrate to N2 gas. To actually determine whether or not nitrate has been reduced, nitrate reduction test is performed with two important reagents viz: Reagent A (sulphanilic acid or nitrate I) Reagent B (dimethyl-alpha-naphthylamine or nitrate II) which makes this test unique.
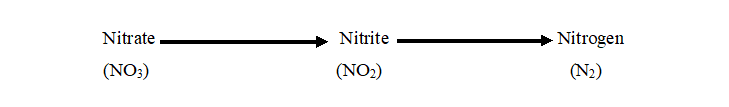
The nitrate reduction test is used to determine whether the test microorganism produces the enzymes nitrate reductase (which breaks down nitrate to nitrite) and nitrite reductase (which breaks down nitrite to nitrogen). Nitrate reductase and nitrite reductase are the two enzymes that catalyze the two reactions involved in converting the starting compound nitrate into the end product nitrogen gas (Figure 1).
PROCEDURE FOR NITRATE REDUCTION TEST
- Prepare nitrate peptone water or broth by constituting peptone water with the addition of 0.2 % potassium nitrate.
- Dispense the prepared medium in test tubes of 10 ml each. This is the nitrate broth.
- Invert Durham tube(s) into each of the test tubes and sterilize in the autoclave.
- After cooling to room temperature, inoculate tube(s) with the test organisms while leaving one tube uninoculated. The uninoculated tube serves as the negative control tube.
- Incubate tubes at 37oC overnight or at 18-24 hrs.
- Add 1 ml each of the two different reagents (A and B) to all the test tubes.
- First observe the tubes for the presence of gas in the Durham tube.
- The tubes turn red if nitrite is present in the medium (i.e. the test bacterium has converted nitrate to nitrite via the production of nitrate reductase). Nitrite in the medium reacts with reagent A and B to form the red colour and this is considered a positive test result.
- Absence of a red colour in the test tubes shows a negative test result. This occurs if NO3– has not been reduced to NO2–, or that NO3– was further reduced to another undetectable compound (e.g. N2 or NH3) after been converted to NO2– (which can be a positive result). However, if the medium does not turn red after the addition of the zinc powder, then the result is called a positive complete. If no red color forms, there was no nitrate to reduce.
- To actually ascertain the reliability of the test result, add zinc compound (Zn) to the test tubes. Presence of red colour after addition of Zn indicates a negative result because the Zn compound will convert any remaining NO3– to NO2–, and this will allow reagents A and B to react with NO2– to form the red colour. Absence of a red colour after adding Zn compound confirms a positive test result, reason being that the test bacterium may have actually reduced nitrate to nitrite and further converted NO2– unnoticeable forms of nitrogen including ammonia and nitrogen (Figure 2). Some bacteria produce both nitrate reductase and nitrite reductase, some produce only nitrate reductase enzyme, while other bacteria produce neither of these enzymes. Pseudomonas aeruginosa and Escherichia coli are examples of bacteria that reduces nitrate to nitrite.

References
Basic laboratory procedures in clinical bacteriology. World Health Organization (WHO), 1991. Available from WHO publications, 1211 Geneva, 27-Switzerland.
Beers M.H., Porter R.S., Jones T.V., Kaplan J.L and Berkwits M (2006). The Merck Manual of Diagnosis and Therapy. Eighteenth edition. Merck & Co., Inc, USA.
Biosafety in Microbiological and Biomedical Laboratories. 5th edition. U.S Department of Health and Human Services. Public Health Service. Center for Disease Control and Prevention. National Institute of Health. HHS Publication No. (CDC) 21-1112.2009.
Cheesbrough M (2010). District Laboratory Practice in Tropical Countries. Part I. 2nd edition. Cambridge University Press, UK.
Cheesbrough M (2010). District Laboratory Practice in Tropical Countries. Part 2. 2nd edition. Cambridge University Press, UK.
Collins C.H, Lyne P.M, Grange J.M and Falkinham J.O (2004). Collins and Lyne’s Microbiological Methods. Eight edition. Arnold publishers, New York, USA.
Disinfection and Sterilization. (1993). Laboratory Biosafety Manual (2nd ed., pp. 60-70). Geneva: WHO.
Garcia L.S (2010). Clinical Microbiology Procedures Handbook. Third edition. American Society of Microbiology Press, USA.
Garcia L.S (2014). Clinical Laboratory Management. First edition. American Society of Microbiology Press, USA.
Fleming, D. O., Richardson, J. H., Tulis, J. I. and Vesley, D. (eds) (1995). Laboratory Safety: Principles and practice. Washington DC: ASM press.
Dubey, R. C. and Maheshwari, D. K. (2004). Practical Microbiology. S.Chand and Company LTD, New Delhi, India.
Gillespie S.H and Bamford K.B (2012). Medical Microbiology and Infection at a glance. 4th edition. Wiley-Blackwell Publishers, UK.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.




