Bacillus anthracis is a mesophilic, Gram-positive, aerobic, catalase-positive, rod-like and spore-forming bacterium that causes anthrax in both humans and animals (e.g. goats, cattle and sheep). Due to their ubiquity in the soil, microbes in the genus Bacillus are often referred to as a normal flora of the soil. B. anthracis is very unique amongst other pathogens in that it was the microbe that was used to establish the first proof that a specific microorganism is responsible for a particular disease.
This remarkable breakthrough about the aetiology of a disease was laid to rest by the work of Robert Koch in 1876 that worked on B. anthracis and concluded both by microscopy and animal inoculation that the microbe was responsible for the disease anthrax. Koch’s postulates which demonstrated for the first time that a specific bacterium is the cause of a specific disease is instrumental in the practice of microbiology and is still applied in medicine even till date. Though other species of Bacillus have recently been implicated in human infections, only B. anthracis and B. cereus (that causes food poisoning) are the most predominant species that are of medical importance, and majorly cause disease in human population.
Anthrax, a highly infectious disease is a zoonotic infection in humans, and the disease can be acquired or transmitted to healthy individuals following contact with infected animals or materials of such animals harbouring the spores of the organism. Spores of B. anthracis are potent bioterrorism agents due to their resistance and prolonged existence which allows them to be airborne for a long period of time.
Bioterrorism isthe deliberate use of microbes (including bacteria, fungi or viruses) or their products (e.g. spores and toxins) to cause death or disease in human populations, plants or animals. Few kilograms of B. anthracis spores can be as deadly as a nuclear bombardment, thus there is a global surveillance mechanism in place by the United Nations (UN) to checkmate and control the illegal usage of biological weapons in warfare.
PATHOGENESIS OF BACILLUS INFECTION
Anthrax is typically the disease of wild and domesticated animals (in particular herbivores). Infections in human population only occurs by chance following human’s contact with infected dead animals, and fluids including excreta from their body. The primary mode of entry of B. anthracis spores into the human body is through inhalation. However, the microbe can also penetrate the body via the skin or GIT. Endospores of B. anthracis, which are the infectious componentof the pathogen are usually formed following nutrient depletion in the surrounding environment of the microbe (e.g. the soil) from where they go on to infect susceptible humans.
Clinically, human anthrax presents in three major clinical forms including: cutaneous anthrax (which is an ulcerative sore on the skin), pulmonary anthrax (which affects the respiratory system) and gastrointestinal anthrax (which affects the GIT), all of which characterize the portal of entry of B. anthracis spores into the human body. Upon invasion, B. anthracis produces two virulence factors (anthrax toxin and poly-D-glutamyl capsule) which enhance its pathogenicity. B. anthracis spores survive attack by phagocytes with the help of its anti-phagocytic capsule (poly-D-glutamyl) and start the formation of exotoxins after spreading to the bloodstream and lymph nodes.
Anthrax toxin is made up of three antigenic components which are: protective antigen (PA) which induces protective antitoxic antibodies in animals, lethal factor (LF) which is essential for the killing effect of anthrax toxin and edema factor (EF) which mediates swelling or oedema on the skin. PA binds to ATRs (which are specific anthrax antigen receptors) on the plasma membranes of their host cell in order to create a channel of entry for EF and LF. The virulence of B. anthracis is attributed to three component of the organism: 1. A poly-D-glutamyl capsule; 2. LF exotoxin; and 3. EF exotoxin.
In pulmonary or inhalation anthrax (Woolsorters’ disease), infection may occur after 1-6 days of inhaling aerosolized B. anthracis spores, and the clinical symptoms may include malaise, shortness of breath, cough, fever, and myalgia. Spores germinate in the lymph nodes en route the respiratory tract including the alveoli, and can reach the meninges where they rarely cause meningitis.
In cutaneous anthrax which usually occur on exposed parts of the body e.g. arms, a local ulceration (oedema) appears on the skin after 3-5 days of B. anthracis spores entry via cut or abrasion. The lesion is usually self-limiting, and heals completely after some days without any scratch mark on the skin.
In gastrointestinal anthrax (which affects the intestines), infections occur following the ingestion of meat from an infected dead animal. Particularly, intestinal anthrax has a high mortality rate, and its symptoms include abdominal pain, bloody vomiting, tenderness, constipation, diarrhea, inflammation and perforation of the GIT. Generally, the anti-phagocytic properties of B. anthracis capsule support the survival of the microbe which enables it to produce sufficient exotoxins to initiate a disease process.
LABORATORY DIAGNOSIS OF BACILLUS INFECTION
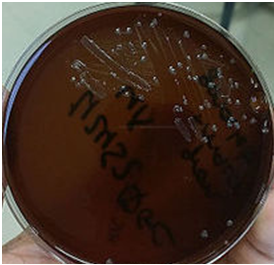
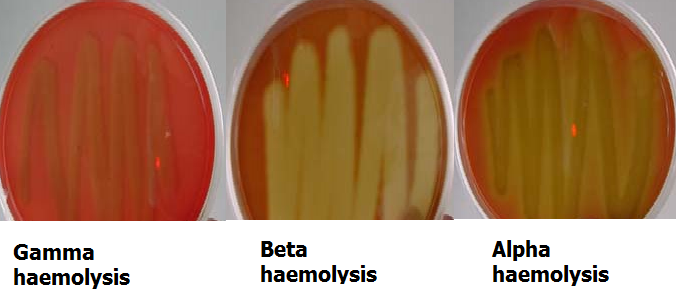
Specimens suspected to containB. anthracis should be regarded as “HIGH RISK”; handled and processed with utmost caution while ensuring that all necessary safety and protection guidelines for such pathogens are dutifully imbibed. Anthrax is diagnosed in the laboratory primarily by the culture of blood, CSF, sputum or skin lesions. B. anthracis spores are very rare in some specimens such as CSF, thus their presence should raise suspicion of anthrax infection. Non-anthrax species of Bacillus such as B. subtilis amongst others are usually easier to cultivate in the laboratory than the pathogenic B. anthracis strain, and these are usually isolated from specimens as environmental contaminants. B. anthracis is a non-motile organism and it does not cause haemolysis on blood agar (Figure 1).
This feature can be used to differentiate it from the saprophytic species of Bacillus which are motile and β-haemolytic. Also, B. anthracis is a large Gram-positive rod in smears, and spores of B. anthracis are usually large with square ends and they are centrally placed or located. Motility in Bacillus species is usually by swarming as is exemplified by Proteus species. Immunoflourescence staining techniques can also be used to detect the microbe from dried smears of specimens including blood, sputum, and fluids from skin lesions.
Animal inoculation can also be used to demonstrate anthrax infection because if cultures of B. anthracis are inoculated intraperitoneally in laboratory animals such as guinea pigs and mice, death will result. Though aerobic, B. anthracis can also be cultured anaerobically; and microscopical examination of sputum, CSF, blood and fluids from skin lesions in smears is often the best method of identifying anthrax bacilli in the laboratory owing to their unique morphological appearance.
Generally, the laboratory diagnosis of anthrax is made on the basis of microscopical identification and growth characteristics of the microbe on culture media especially the sheep’s blood agar medium. Further confirmatory tests on the suspect pathogen can also be ascertained by sending samples to reference laboratory in order to confirm or out rule any possible form of bioterrorism.
IMMUNITY TO BACILLUS INFECTION
Immunity against anthrax infection or re-infection is complex and they still remain arcane. Anthrax infection confers varying immunity in infected animals, and those surviving a natural infection by B. anthracis are normally immune to re-infection. Any effective vaccine against anthrax must be able to elicit antibodies against the -D-glutamyl capsule and toxins (LF and EF) of B. anthracis since these factors are critical in propagating the virulence and pathogenicity of the pathogen.
However, vaccines comprising live attenuated spore and avirulent non-encapsulated strain of B. anthracis have been used in some parts of the world including the U.S. and Russia in human population with some proven efficacy. Immunization against anthrax infection usually consists of three subcutaneous injections which are given separately in two weeks. This is then followed by three additional subcutaneous injections which are given at 6, 12, and 18 months. But protective level of immunity against the infection is usually attained via annual booster injections of the vaccine.
Anthrax vaccine for humans is contraindicated for pregnant women, and it should be administered to persons age 18 and above especially those in contact with infected animals and their products such blood, hides and skin, and hairs. Passive anthrax vaccines are usually more effective especially in suspected cases of bioterrorism because such vaccines elicit rapid antibody production in vivo and confer immunity faster than active vaccines that usually require many administrations over a long period of time to elicit a protective immunity.
TREATMENT OF BACILLUS INFECTION
Early treatment using antibiotics is critical in B. anthracis infection. B. anthracis is sensitive to cotrimoxazole, tetracyclines, penicillins, erythromycin, aminoglycosides and fluoroquinolones. A handful of these antibiotics are effective in treating the 3 clinical forms of anthrax (pulmonary, gastrointestinal and cutaneous) especially if administered before symptoms appear or immediately after exposure to B. anthracis spores.
Co-administration of antibiotics and passive vaccine in the treatment of anthrax has shown to enhance therapy using antibiotics. Anthrax is usually very difficult to treat especially when the endospores of the pathogen spreads and disseminates to other vital tissues and organs of the body.
Early symptoms of the diseases including flu-like signs should be properly diagnosed, and once B. anthracis infection has been confirmed, treatment should commence immediately using any of the above antibiotics in order to prevent death due to the rapid progression of the infection in the body.
PREVENTION AND CONTROL OF BACILLUS INFECTION
Anthrax is a worldwide infection, and the resistant nature of B. anthracis endospores can encourage the transmission and spread of the microbe from one geographical location to another. Spores of B. anthracis are mostly found in agricultural regions of the world especially in places where herbivores are reared, and people in contact with these animals and their products are mostly at risk of acquiring the pathogen.
The vaccination of animals especially herbivores and livestock is important in the control of B. anthracis infection. People in direct or indirect contact with these animals should also be regularly immunized against the pathogen in order to avoid spread and transmission to susceptible individuals. Spores of B. anthracis in the soil can become aerosolized and go on to infect susceptible humans to cause inhalation anthrax, and this usually occurs in places where the soil is contaminated with cadavers and remains of infected dead animals.
Humans should minimize as much as possible their contacts with potentially infected animals and their remains. Protective clothing should be used by people handling potentially infected animals. Remains of infected dead animals should be burned and buried very deep in the soil. In cases of bioterrorism, only already vaccinated armed forces personnel can adequately respond to any biological attack involving B. anthracis, thus it is vital that that men and women in the military and other armed forces be properly vaccinated before hand in order to better prepare them against such attack should it happen.
OTHER SPECIES OF BACILLUS
- Bacillus cereus: Bacillus cereus causes food poisoning in humans. B. cereus can also cause opportunistic infections in humans.
- Bacillus thuringiensis: B. thuringiensis is an important insect pathogen, and it is widely used as commercial insecticides to control pest attack in plants.
- Bacillus subtilis: B. subtilis is used in antibiotic production.
- Bacillus popilliae: B. popilliae is used as commercial insecticides to control pest attack in plants.
- Bacillus sphaericus: B. sphaericus is used as commercial insecticides to control pest attack in plants.
- Bacillus larvae: B. larvae is used as commercial insecticides to control pest attack in plants.
- Bacillus lentimorbus: B. lentimorbus is used as commercial insecticides to control pest attack in plants.
Bacillus species used as commercial insecticides (e.g. B. thuringiensis) produce substantial amount of insect larvicides (i.e. chemical substances that destroy the larval stages of insects) that can be used in agricultural purposes to control pest attack on crops and vegetations while ensuring food security.
References
Brooks G.F., Butel J.S and Morse S.A (2004). Medical Microbiology, 23rd edition. McGraw Hill Publishers. USA. Pp. 248-260.
Madigan M.T., Martinko J.M., Dunlap P.V and Clark D.P (2009). Brock Biology of microorganisms. 12th edition. Pearson Benjamin Cummings Publishers. USA. Pp.795-796.
Prescott L.M., Harley J.P and Klein D.A (2005). Microbiology. 6th ed. McGraw Hill Publishers, USA. Pp. 296-299.
Ryan K, Ray C.G, Ahmed N, Drew W.L and Plorde J (2010). Sherris Medical Microbiology. Fifth edition. McGraw-Hill Publishers, USA.
Singleton P and Sainsbury D (1995). Dictionary of microbiology and molecular biology, 3rd ed. New York: John Wiley and Sons.
Talaro, Kathleen P (2005). Foundations in Microbiology. 5th edition. McGraw-Hill Companies Inc., New York, USA.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.