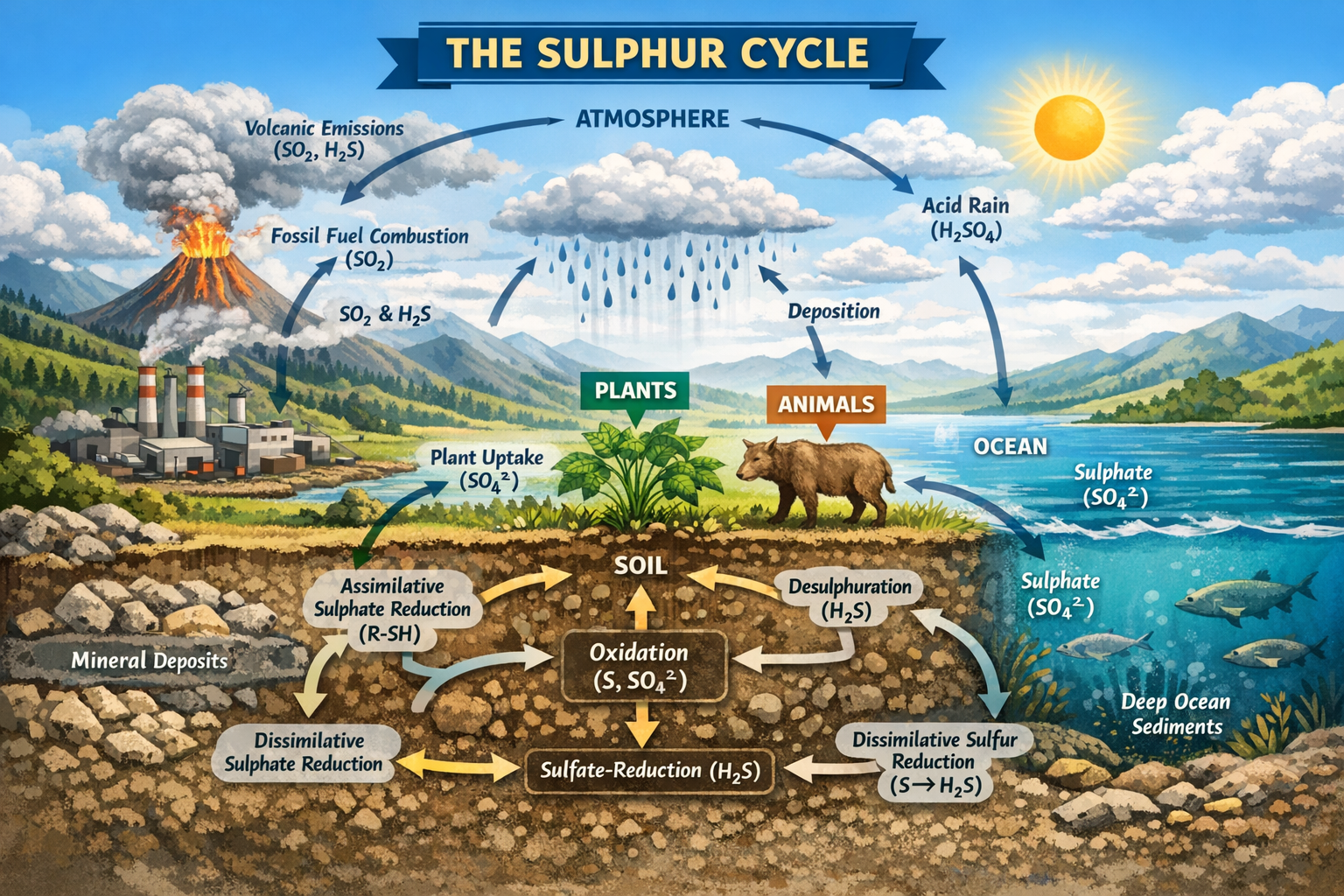
Active air monitoring also involve the use of settle plates or sedimentation culture plates (as is applicable in passive air sampling) and contact plates for the monitoring of air quality. It involves extracting a set volume of air within a given environment into a calibrated sampler which is then passed onto the surface on an agar plate. The agar culture plates are then incubated at ambient temperature to allow the growth of colonies of airborne microbes including moulds and bacteria.
There is an increasing need for effective air monitoring in all clean areas with filtered air where airborne microorganisms may contaminate or affect industrial products and processes such as in biopharmaceutical companies, hospitals and food industries. Active air monitoring with contact plates requires the use of a microbiological air sampler to physically draw a certain, pre-determined volume of air and pass it over the agar culture plate on the air sampling device also known as particle collection devices.
As aforementioned, the exposed culture media plate is removed from the microbiological air sampler and directly incubated at ambient temperature for microbial growth. The resulting colonies which give an estimate of the number of colony forming units in the sampled air are counted and reported quantitatively per taxon identified in Colony Forming Units per cubic metre of air (CFU/m³). Contact plates are usually used in the active air monitoring techniques. The contact plates are used to measure microbial contamination of work surfaces in manufacturing or clean rooms of food industries and pharmaceutical companies.
The surface of the medium is pressed against a flat sampling surface to pick up any microorganisms that may have settled onto the location by operator contact or from the environment. Active air monitoring requires the use of a microbiological air sampler to physically draw a known volume of air over, or through, a particle collection device (Figure 1). There are various types of these particle collection devices but only the impingers particle collection device and the impactor particle collection device or samplers are highlighted below:

- Impingers or impinger samplers use a liquid medium for particle collection during active air sampling. In the use of this device, the sampled air is drawn by a suction pump through a narrow inlet tube into a small flask containing the collection culture medium. This accelerates the air towards the surface of the collection medium and the flow rate is determined by the diameter of the inlet tube.
When the air hits the surface of the liquid, it changes direction abruptly and any suspended particles are impinged into the collection culture media. The collection culture medium is then cultured to enumerate viable microorganisms once the sampling is complete. The result of this technique is quantitative since the sample volume can be calculated using the flow rate and sampling time. The use of impingers for active air monitoring has some disadvantages associated with its use. Most impingers are usually made of glass, and this is undesirable in food and pharmaceutical production sites. The impingement of the sampled air into culture media may also damage some microbial cells and thus affect the viability of the result. Also, the longtime of sampling may allow some cells to multiply in the liquid collection medium.
- Impactors or impactor samplers use a solid or adhesive culture medium (e.g. agar) for particle collection unlike the impinger devices that use liquid culture medium in active air sampling technique. They are commonly used for active air sampling because of their convenience. In a typical impactor sampler, air is drawn into a sampling head by a pump or fan and accelerated, usually through a perforated plate (sieve samplers), or through a narrow slit (slit samplers). This produces laminar air flow onto the collection surface that is usually a standard agar culture plate or contact plate filled with a suitable agar medium that favour microbial growth.
The velocity of the air is determined by the diameter of the holes in sieve samplers and the width of the slit in slit samplers. When the air hits the collection surface of the culture medium, it makes a tangential change of direction and any suspended particles are thrown out by the force of inertia, thus allowing the particles to impact onto the collection surface. When the correct volume of air has been passed through the sampling head, the agar culture plate is removed and incubated at ambient temperature for microbial growth. After incubation, the visible microbial growth (colonies) on the agar plate is counted and reported as CFU/m³. This gives a direct quantitative estimate of the number of colony forming units in the sampled air.
The impactor/impaction samplers have several benefits over the impinger samplers. It is convenient to use and it makes use of pre-poured solid culture media plates. They are also able to handle higher flow rates and the large sample volumes necessary to monitor air quality in clean rooms where the number of microbes present is likely to be very low. Proper care must be taken not to allow agar plates to remain in the sampler heads for too long, or the culture medium may dry out and deteriorate. Microbial cells may also be damaged by mechanical stress during the sampling process and lose viability. Impaction samplers offer benefits in terms of convenience and pre-poured, gamma-irradiated contact plates and standard Petri dishes from specialist suppliers can be used with them to minimize the risk of contamination and variation.
References
Arora D.R (2004). Quality assurance in microbiology. Indian J Med Microbiol, 22:81-86.
Ashutosh Kar (2008). Pharmaceutical Microbiology, 1st edition. New Age International Publishers: New Delhi, India.
Axelsen P.H (2002). Essentials of antimicrobial pharmacology. Humana Press, Totowa, New Jersey, USA. Al-Jasser A.M (2006). Extended – Spectrum Beta – Lactamases (ESBLs): A Global Problem. Kuwait Medical Journal, 38(3):171-185.
Bisht R., Katiyar A., Singh R and Mittal P (2009). Antibiotic Resistance – A Global Issue of Concern. Asian Journal of Pharmaceutical and Clinical Research, 2 (2):34-39.
Block S.S (2001). Disinfection, sterilization and preservation. 5th edition. Lippincott Williams & Wilkins, Philadelphia and London.
Joslyn, L. J. (2000). Sterilization by Heat. In S. S. Block (Ed.), Disinfection, Sterilization, and Preservation (5th ed., pp. 695-728). Philadelphia, USA: Lippincott Williams and Wilkins.
Nally J.D (Ed.) (2007). Good manufacturing practices for pharmaceuticals. Sixth edition. Informa Healthcare USA, Inc, New York.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.