Hazard Analysis and Critical Control Point (HACCP) is an internationally recognized, science-based food safety management system applied throughout the food production and processing chain to ensure food safety, hygiene, and overall product quality. Originally developed to enhance the microbiological safety of foods for space missions, HACCP has evolved into a globally adopted preventive framework endorsed by regulatory authorities and international bodies such as the Food and Agriculture Organization (FAO) and the World Health Organization (WHO). It is now widely implemented across primary production, manufacturing, packaging, distribution, and retail sectors within the food industry.
HACCP functions as a systematic quality assurance methodology that identifies, evaluates, and controls hazards that are reasonably likely to occur at specific stages of the food production process. These hazards are generally categorized into three principal classes: biological hazards (e.g., pathogenic microorganisms such as bacteria, viruses, and parasites), chemical hazards (e.g., pesticide residues, cleaning agents, allergens, or toxins), and physical hazards (e.g., foreign objects such as metal fragments, glass, or plastic). Rather than relying solely on end-product inspection and testing, HACCP emphasizes preventive control measures embedded within the process itself, thereby shifting the focus from reactive correction to proactive risk mitigation.
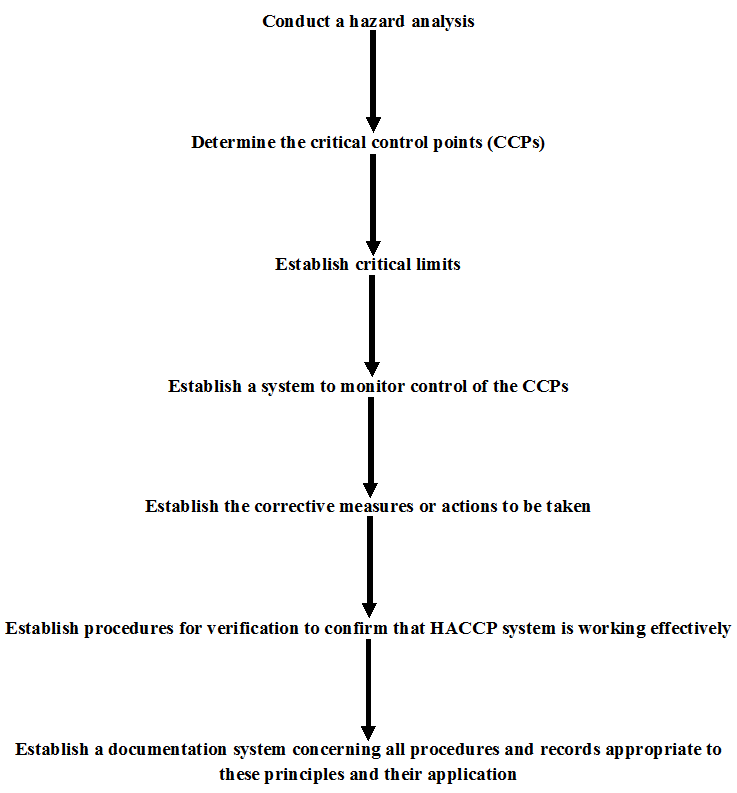
The HACCP system is structured around seven core principles: conducting a hazard analysis; determining critical control points (CCPs); establishing critical limits for each CCP; implementing monitoring procedures; defining corrective actions; verifying that the system is functioning effectively; and maintaining comprehensive documentation and record-keeping. Through these principles, food business operators can systematically assess potential risks inherent in raw materials, processing steps, equipment, personnel practices, storage conditions, and distribution channels.
By identifying points in the production line where control is essential to prevent, eliminate, or reduce hazards to acceptable levels, HACCP enables organizations to implement sustainable and evidence-based control measures. These may include temperature control, sanitation protocols, supplier verification, allergen management, and equipment maintenance programs. The result is a robust food safety management system that enhances consumer protection, supports regulatory compliance, reduces product recalls, and strengthens brand credibility. Overall, HACCP represents a cornerstone of modern food safety governance and a critical tool for ensuring the consistent production of safe, high-quality food products.
Hazard Analysis and Critical Control Point (HACCP) is a systematic, science-based food safety management system built on seven core principles that collectively establish the foundation for maintaining hygienic conditions and regulatory compliance throughout food production processes (Figure 1). These principles include hazard analysis, identification of critical control points (CCPs), establishment of critical limits, monitoring procedures, corrective actions, verification activities, and documentation systems. The HACCP framework is designed to proactively identify biological, chemical, and physical hazards that may arise at any stage of the food supply chain—from raw material sourcing to processing, storage, and distribution. Once identified, these hazards are rigorously evaluated in terms of likelihood and severity to determine their potential impact on consumer health. Appropriate control measures are then implemented at designated CCPs to prevent, eliminate, or reduce hazards to acceptable levels. Through continuous monitoring and verification, HACCP ensures effective hazard control and sustained food safety performance.
Food processing companies are obligated to implement the principles of Hazard Analysis and Critical Control Points (HACCP) at every stage of production to systematically identify, evaluate, and control potential hazards that could compromise the safety of their final products. HACCP is a preventive, science-based framework that focuses on hazard identification and risk management rather than reliance on end-product testing alone. By embedding HACCP into their operational architecture from raw material procurement to distribution – food manufacturers can eliminate or reduce biological, chemical, and physical hazards to acceptable levels before products reach consumers.
Although governmental authorities bear primary responsibility for safeguarding public health through regulatory oversight, inspection, and enforcement, food safety is not solely a regulatory function. Governments establish statutory standards, conduct audits, and monitor compliance to ensure that food processing facilities operate within legal and public health parameters. However, regulatory surveillance cannot substitute for robust internal control systems. The ultimate responsibility for producing safe, wholesome, and hygienic food rests with the food industry itself. Manufacturers must adopt a proactive compliance posture, ensuring that their operations consistently meet or exceed regulatory benchmarks and that their products pose no health risk to consumers.
Food industries that prioritize safety and hygiene treat HACCP not as a procedural formality but as an integral component of their quality management systems. They integrate HACCP principles with Good Manufacturing Practices (GMPs), Sanitation Standard Operating Procedures (SSOPs), and other prerequisite programs to create a comprehensive food safety management framework. GMPs provide the foundational operational and environmental conditions necessary for hygienic production—covering facility design, equipment maintenance, water quality, pest control, personnel hygiene, and sanitation protocols. HACCP builds upon these prerequisites by introducing structured hazard analysis and the identification of Critical Control Points (CCPs) where preventive or corrective measures can be applied effectively.
The seven principles of HACCP provide a systematic methodology for ensuring food safety. First, a thorough hazard analysis is conducted to identify potential biological hazards (such as pathogenic microorganisms), chemical hazards (including allergens, toxins, or residues), and physical hazards (such as foreign objects). Second, CCPs are determined – these are stages in the process where control is essential to prevent, eliminate, or reduce hazards to acceptable levels. Third, critical limits are established for each CCP, defining measurable parameters such as time, temperature, pH, or moisture content. Fourth, monitoring procedures are implemented to ensure that each CCP remains within its critical limits. Fifth, corrective actions are defined to address deviations promptly. Sixth, verification procedures are carried out to confirm that the HACCP system functions effectively. Finally, comprehensive documentation and record-keeping ensure traceability, accountability, and regulatory compliance.
Personnel competence is central to the effective implementation of HACCP. Employees involved in production, quality assurance, and laboratory testing must be adequately trained in food hygiene, hazard recognition, and control procedures. A well-designed HACCP plan is only as effective as the people responsible for executing it. Continuous professional development, internal audits, and management review processes are therefore essential to maintain operational integrity and foster a culture of food safety within the organization.
Testing of end products, while important, serves primarily as a verification tool rather than the primary control strategy. Reliance solely on finished product inspection is insufficient because hazards may not be uniformly distributed, and contamination events can be sporadic. Instead, process control through validated CCP monitoring and preventive measures – offers a more reliable assurance of safety. Quality control systems must be integrated with HACCP to ensure that deviations are detected early and rectified before products enter the market.
A hygienic production environment is equally critical. Environmental monitoring programs, effective sanitation regimes, controlled personnel movement, and proper waste management collectively minimize cross-contamination risks. Infrastructure design should support hygienic zoning and prevent recontamination after critical interventions such as thermal processing. Maintenance of equipment, calibration of instruments, and validation of cleaning procedures further strengthen hazard control mechanisms.
Ultimately, food must be hygienically safe to gain consumer acceptance and protect public health. Consumers expect that foods available in the marketplace are free from contaminants and will not cause illness or injury when consumed under normal conditions of use. By integrating HACCP with GMPs and fostering a culture of accountability and continuous improvement, food processing companies can meet this expectation. The synergy between regulatory oversight and industry self-regulation ensures that food safety is maintained not merely as a legal obligation but as an ethical and professional imperative.
Most microbiological assays conducted in food production facilities are designed primarily to detect contamination by non-specific indicator organisms rather than to identify particular pathogens at the outset. In routine quality control laboratories, these tests often target broad physiological or metabolic traits that signify microbial growth. A common example is the detection of gas production during carbohydrate fermentation, which serves as an indirect marker for the presence of coliforms and other fermentative bacteria. Gas accumulation in selective broths or Durham tubes, along with changes in pH, turbidity, or colorimetric indicators, provides rapid evidence of microbial contamination without requiring immediate species-level identification. Such indicator-based testing is widely adopted because it is cost-effective, scalable, and suitable for high-throughput screening in industrial settings.
Microbiological methods in food industries therefore frequently rely on the detection of metabolic by-products—organic acids, gases (e.g., CO₂ and H₂), hydrogen sulfide, or enzymatic activities that function as proxies for microbial presence and proliferation. These approaches are particularly useful for monitoring hygiene status, process control, and compliance with regulatory microbiological criteria. However, when greater specificity is required – especially for the detection of Gram-negative bacteria more targeted assays may be employed. One such method is the Limulus Amebocyte Lysate test, which detects lipopolysaccharides (LPS), the major endotoxin component of the outer membrane of Gram-negative organisms. The presence of LPS in a food sample indicates contamination by Gram-negative bacteria, even if viable cells are no longer detectable. Depending on the format (gel-clot, turbidimetric, or chromogenic), the assay can provide qualitative or quantitative measurements of endotoxin levels, thereby enhancing sensitivity and specificity compared to conventional culture-based tests.
Molecular diagnostic tools have further expanded the analytical capacity of food microbiology laboratories. Polymerase chain reaction (PCR) technology enables the rapid and highly specific detection of microbial DNA directly from food matrices. When species-specific or strain-specific primers are available, PCR assays can confirm the presence of particular pathogens with high analytical sensitivity, often within hours. Variants such as real-time quantitative PCR (qPCR) additionally allow for the estimation of microbial load. These molecular approaches reduce detection time relative to traditional culture techniques and are especially valuable for fastidious or slow-growing organisms.
Beyond laboratory testing, microbiological safety in food production is underpinned by preventive risk management systems such as Hazard Analysis and Critical Control Points (HACCP). HACCP involves a systematic identification of biological, chemical, and physical hazards within the production process, followed by the establishment of critical control points (CCPs) where risks can be prevented, eliminated, or reduced to acceptable levels. By integrating routine microbiological testing with structured process control, food manufacturers can ensure product safety, regulatory compliance, and consumer protection.
In quality and safety management systems, particularly under Hazard Analysis and Critical Control Point (HACCP) frameworks, the term “hazard” denotes any biological, chemical, or physical factor with the potential to compromise the integrity of the production process or the safety and quality of the final product. If not identified and mitigated at an early stage, such hazards can propagate through the processing chain, resulting in product contamination, regulatory non-compliance, economic losses, and potential risks to consumer health. Effective hazard control therefore depends on a systematic and comprehensive hazard analysis conducted prior to, and throughout, production.
A thorough hazard assessment entails the structured evaluation of all known and reasonably foreseeable risks associated with the manufacturing process. This includes hazards intrinsic to raw materials (e.g., microbial load, chemical residues, foreign bodies), those introduced through processing steps (e.g., cross-contamination, equipment malfunction), and those linked to personnel practices (e.g., inadequate hygiene, improper handling techniques). Environmental factors such as air quality, water sources, and surface sanitation must also be scrutinized. The objective of this assessment is to determine the likelihood and severity of each hazard, thereby enabling prioritization of control measures based on risk characterization.
Following hazard identification and risk evaluation, the next critical step is the determination of Critical Control Points (CCPs), as illustrated in Figure 1. CCPs are defined as specific stages in the production process at which control can be applied and is essential to prevent, eliminate, or reduce a food safety hazard to an acceptable level. These points represent operational thresholds where failure to exercise adequate control would result in an unacceptable safety risk. The selection of CCPs is typically guided by decision trees or structured risk-assessment tools to ensure objectivity and consistency.
At each identified CCP, measurable critical limits must be established. These limits may include parameters such as temperature, time, pH, moisture content, or microbial counts, depending on the nature of the hazard being controlled. Routine and systematic monitoring at CCPs is indispensable. Monitoring activities should be designed to detect deviations in real time or near real time, enabling immediate corrective actions before compromised products proceed further along the production line.
Microbiological assessment constitutes a central component of monitoring at CCPs, particularly where biological hazards are of primary concern. This involves the targeted detection and quantification of specific microorganisms known to cause spoilage or contamination. Indicator organisms and, where appropriate, pathogenic species are assayed using validated analytical methods. Timely reporting of microbiological results is essential to facilitate rapid intervention, including process adjustments, product segregation, or temporary production shutdowns where necessary. Such proactive monitoring ensures process control, safeguards product quality, and upholds regulatory and consumer safety standards.
References
Bushell M.E (1998). Application of the principles of industrial microbiology to biotechnology (ed. Wiseman, A.) Chapman and Hall, New York.
Byong H. Lee (2015). Fundamentals of Food Biotechnology. Second edition. Wiley-Blackwell, New Jersey, United States.
Clark D.P and Pazdernik N (2010). Biotechnology. First edition. Elsevier Science and Technology Books, Amsterdam, Netherlands.
Farida A.A (2012). Dairy Microbiology. First edition. Random Publications. New Delhi, India.
Frazier W.C, Westhoff D.C and Vanitha N.M (2014). Food Microbiology. Fifth edition. McGraw-Hill Education (India) Private Limited, New Delhi, India.
Guidebook for the preparation of HACCP plans (1999). Washington, DC, United States Department of Agriculture Food Safety and Inspection Service. Accessed on 20th February, 2015 from: http://www.fsis.usda.gov
Hayes P.R, Forsythe S.J (1999). Food Hygiene, Microbiology and HACCP. 3rd edition. Elsevier Science, London.
Hussaini Anthony Makun (2013). Mycotoxin and food safety in developing countries. InTech Publishers, Rijeka, Croatia. Pp. 77-100.
Jay J.M (2005). Modern Food Microbiology. Fourth edition. Chapman and Hall Inc, New York, USA.
Lightfoot N.F and Maier E.A (1998). Microbiological Analysis of Food and Water. Guidelines for Quality Assurance. Elsevier, Amsterdam.
Nduka Okafor (2007). Modern industrial microbiology and biotechnology. First edition. Science Publishers, New Hampshire, USA. Roberts D and Greenwood M (2003). Practical Food Microbiology. Third edition. Blackwell publishing Inc, USA.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.