The history of antibiotics represents one of the most remarkable achievements in the history of science and medicine. Beginning with early observations of antimicrobial substances and culminating in the discovery of penicillin and subsequent antibiotics, these drugs have transformed the treatment of infectious diseases and saved millions of lives.
Antibiotics are among the most transformative discoveries in the history of medicine. Their introduction fundamentally changed the treatment of infectious diseases and significantly reduced mortality associated with bacterial infections. Before antibiotics became available, infections such as pneumonia, tuberculosis, septicemia, and wound infections frequently resulted in death. Minor injuries could progress into life-threatening infections, and surgical procedures carried substantial risks due to bacterial contamination.
The discovery and development of antibiotics marked the beginning of what is commonly referred to as the “antibiotic era.” This period, which began in the early twentieth century, saw rapid scientific progress in microbiology, pharmacology, and industrial biotechnology. Antibiotics enabled physicians to treat infections that were previously considered untreatable and played a major role in enabling modern medical advances, including complex surgeries, organ transplantation, chemotherapy, and intensive care medicine.
The development of antibiotics did not occur through a single discovery but rather through the combined contributions of several scientists across multiple disciplines. Pioneers such as Alexander Fleming, Howard Florey, Ernst Chain, Selman Waksman, Albert Schatz, and Paul Ehrlich laid the foundations for modern antimicrobial therapy. The collaborative efforts of these scientists played crucial roles in establishing the foundations of antimicrobial therapy. Their discoveries ushered in the antibiotic era and revolutionized modern medicine. Their work ranged from early observations of antimicrobial compounds produced by microorganisms to the development of methods for large-scale antibiotic production.
Today, antibiotics remain essential tools in medicine, agriculture, and veterinary practice. However, the widespread and sometimes inappropriate use of antibiotics has contributed to the emergence of antimicrobial resistance (AMR), which now represents one of the most significant global public health challenges. Understanding the historical development of antibiotics therefore provides important insight into both their remarkable benefits and the challenges associated with their long-term use.
However, the continued effectiveness of antibiotics is threatened by the growing problem of antimicrobial resistance. As bacteria evolve mechanisms to evade existing drugs, the development of new antibiotics and the responsible use of existing ones have become critical priorities.
Understanding the historical development of antibiotics provides valuable insight into both their life-saving potential and the challenges that accompany their widespread use. As the world faces increasing threats from resistant pathogens, the lessons learned from the history of antibiotics will remain essential in guiding future strategies for combating infectious diseases.
Early Observations of Antimicrobial Effects
Although the scientific discovery of antibiotics occurred in the twentieth century, the use of natural substances with antimicrobial properties dates back thousands of years. Ancient civilizations used plant extracts, molds, and fermented materials to treat infections and wounds. For example, historical records suggest that ancient Egyptians, Greeks, and Chinese applied moldy bread or plant-based preparations to infected wounds in an attempt to promote healing. While these practices were not based on microbiological knowledge, they represent early empirical recognition of antimicrobial activity in natural substances.
The scientific foundation for understanding infectious diseases began to emerge in the nineteenth century with the development of the germ theory of disease. Scientists such as Louis Pasteur and Robert Koch demonstrated that microorganisms were responsible for many infectious diseases. Their work provided the conceptual framework necessary for the development of antimicrobial therapies.
During this period, researchers began to explore chemical compounds that could selectively kill microorganisms without harming the host organism. One of the most influential scientists in this area was Paul Ehrlich, a German physician and scientist who introduced the concept of “selective toxicity.” Ehrlich proposed that it should be possible to design chemicals that specifically target microbial pathogens while leaving human cells unharmed.
In the early twentieth century, Ehrlich and his colleagues developed arsphenamine (Salvarsan), which was used to treat syphilis caused by the bacterium Treponema pallidum. Introduced in 1910, Salvarsan is often considered the first modern antimicrobial drug. Although it was not technically classified as an antibiotic, its development demonstrated that chemical compounds could be used to treat bacterial infections effectively.
Selective Toxicity
Selective toxicity refers to the capacity of an antimicrobial agent to specifically target and inhibit microbial pathogens while causing minimal harm to the host organism. This concept is fundamental to the development and effective use of antibiotics in clinical medicine. It is based on the biological and structural differences between microbial cells and human cells. For example, many antibiotics target bacterial structures such as the peptidoglycan cell wall, bacterial 70S ribosomes, or metabolic pathways like folic acid synthesis, which are absent or significantly different in human cells. By exploiting these differences, antibiotics can disrupt essential bacterial processes such as cell wall formation, protein synthesis, or DNA replication, ultimately leading to bacterial death or growth inhibition. The principle of selective toxicity allows antibiotics to effectively eliminate pathogenic microorganisms while reducing potential damage to host tissues, thereby making them safe and effective therapeutic agents for treating bacterial infections.
The Discovery of Penicillin
The most famous and historically significant discovery in the history of antibiotics occurred in 1928 when the Scottish bacteriologist Alexander Fleming observed the antibacterial effects of a mold belonging to the genus Penicillium. At the time, Fleming was working at St. Mary’s Hospital in London, where he was studying cultures of Staphylococcus aureus, a bacterium responsible for a wide range of human infections including wound infections, pneumonia, and septicemia. His research focused on identifying substances capable of inhibiting bacterial growth, an area of increasing interest as scientists sought effective treatments for infectious diseases.
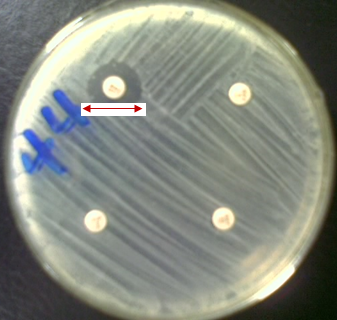
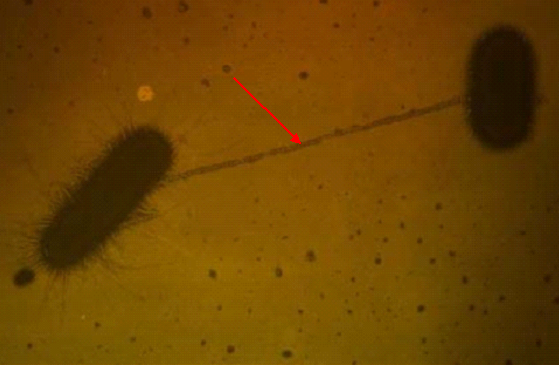
According to historical accounts, Fleming returned from a short vacation to find that one of his Petri dishes had been accidentally contaminated by a mold. Instead of discarding the contaminated culture, he carefully examined it and noticed an unusual phenomenon. The bacterial colonies surrounding the mold had been destroyed, leaving a distinct clear zone where bacterial growth was completely inhibited. This clear area, later described as a “zone of inhibition,” suggested that the mold was producing a substance capable of killing or suppressing nearby bacteria.
Recognizing the potential significance of this observation, Fleming conducted further experiments to investigate the antibacterial activity of the mold. He demonstrated that the substance produced by the mold was particularly effective against several Gram-positive bacteria, including Staphylococcus and Streptococcus species. These findings indicated that the mold secreted a diffusible compound with potent antimicrobial properties.
Subsequent analysis revealed that the mold responsible for the antibacterial activity belonged to the species Penicillium notatum. Fleming named the antibacterial substance produced by the mold penicillin, marking the first identification of a naturally occurring antibiotic capable of inhibiting bacterial growth. In 1929, he published his findings in the British Journal of Experimental Pathology, describing the antibacterial properties of penicillin and highlighting its potential therapeutic applications.
Despite the groundbreaking nature of this discovery, Fleming faced significant technical challenges. Penicillin was highly unstable and difficult to isolate in a purified form, making it unsuitable for immediate clinical use. As a result, although Fleming recognized its medical potential, the practical development of penicillin remained limited for more than a decade. It was not until the late 1930s and early 1940s that other researchers successfully purified and produced penicillin on a scale suitable for therapeutic application, ultimately transforming Fleming’s initial observation into one of the most important medical breakthroughs of the twentieth century.
Isolation and Development of Penicillin
The transformation of penicillin from a simple laboratory observation into a clinically useful therapeutic agent occurred more than a decade after its initial discovery, largely through the pioneering efforts of a multidisciplinary research team at the University of Oxford. This team was led by the Australian pathologist Howard Florey and the German-born biochemist Ernst Boris Chain. Their collaboration marked a critical turning point in the history of antimicrobial chemotherapy, as they successfully translated the earlier observations of Alexander Fleming into a viable medical treatment.
In 1939, Florey, Chain, and their colleagues initiated a systematic research program aimed at investigating naturally occurring antimicrobial substances produced by microorganisms. During their investigations, they revisited Fleming’s 1929 publication describing the antibacterial properties of penicillin produced by the mold Penicillium. Recognizing the therapeutic potential of this compound, the Oxford team began developing methods to isolate, purify, and stabilize penicillin for experimental use. This work involved significant biochemical and microbiological innovation, as penicillin was highly unstable and difficult to extract in sufficient quantities using the techniques available at the time.
Through extensive laboratory experimentation, the researchers succeeded in producing crude penicillin extracts and demonstrated that the compound exhibited strong antibacterial activity against a range of pathogenic bacteria, particularly Gram-positive organisms such as Staphylococcus and Streptococcus species. Subsequent animal experiments provided compelling evidence of its therapeutic potential. In one landmark study, mice infected with lethal doses of bacteria survived when treated with penicillin, whereas untreated control animals succumbed to infection. These findings provided the first clear demonstration of penicillin’s effectiveness in treating bacterial infections in vivo.
Encouraged by these results, the Oxford group proceeded to conduct the first clinical trials of penicillin in 1941. Early treatments were administered to patients suffering from severe bacterial infections, including septicemia, abscesses, and wound infections. The clinical outcomes were highly promising, with several patients showing rapid improvement following penicillin therapy. However, these early trials were constrained by the extremely limited supply of the drug. Producing penicillin required large quantities of mold cultures, and extraction yields were very low.
Consequently, the success of these early trials highlighted the urgent need for methods capable of producing penicillin on a much larger scale. This realization ultimately stimulated collaborative efforts between academic researchers, government agencies, and pharmaceutical companies to develop industrial-scale fermentation techniques, paving the way for the widespread availability of penicillin in the following years.
Industrial Production of Penicillin
The outbreak of the World War II significantly accelerated global efforts to produce penicillin on an industrial scale. During the early years of the war, bacterial infections were a major cause of death among wounded soldiers, often surpassing fatalities from the initial injuries themselves. Recognizing the enormous therapeutic potential of penicillin in preventing and treating wound infections, governments in both the United Kingdom and the United States prioritized the rapid development of large-scale production methods. Collaborative initiatives were established between academic researchers, government agencies, and pharmaceutical companies to transform penicillin from a laboratory discovery into a widely available medical treatment.
Early penicillin production faced several challenges, including low yields from naturally occurring strains of the mold Penicillium. Scientists therefore began searching for improved strains capable of producing larger quantities of the antibiotic. Through extensive screening and mutagenesis experiments, researchers identified high yielding strains of Penicillium that significantly increased production efficiency. One particularly productive strain was discovered on a moldy cantaloupe in a market in Peoria, Illinois, which later became instrumental in large-scale manufacturing.
At the same time, advances in industrial microbiology and biochemical engineering led to the development of deep-tank fermentation techniques. These methods involved cultivating Penicillium in large, aerated fermenters containing nutrient-rich media. The introduction of controlled fermentation conditions including optimized temperature, pH, aeration, and nutrient supply allowed microorganisms to grow rapidly and produce penicillin in much greater quantities than previously possible. Pharmaceutical companies such as Pfizer and Merck & Co. played key roles in refining these fermentation processes and scaling them up for industrial production.
By 1943, large-scale penicillin production had begun in the United States, enabling the drug to be distributed widely to Allied military forces. The availability of penicillin dramatically improved the treatment of battlefield infections, reducing mortality rates and preventing complications such as septicemia and gangrene. Penicillin quickly became known as a “miracle drug” due to its remarkable effectiveness and relatively low toxicity.
Following the end of the war, industrial production continued to expand, making penicillin widely accessible for civilian healthcare. Its success marked the beginning of the modern antibiotic era and stimulated extensive research into the discovery and development of additional antimicrobial agents. In recognition of their groundbreaking contributions to the discovery and development of penicillin, Alexander Fleming, Howard Florey, and Ernst Boris Chain were jointly awarded the Nobel Prize in Physiology or Medicine in 1945.
Mechanism of Action of Penicillin
Penicillin belongs to a class of antibiotics known as β-lactams, which exert their antibacterial effects by interfering with the synthesis of the bacterial cell wall. The bacterial cell wall contains a structural component known as peptidoglycan, which provides mechanical strength and protects the cell from osmotic pressure.
β-lactam antibiotics inhibit enzymes known as penicillin-binding proteins (PBPs) that are involved in the cross-linking of peptidoglycan strands during cell wall synthesis. When these enzymes are inhibited, the bacterial cell wall becomes structurally weakened.
As a result, the bacterial cell becomes susceptible to osmotic pressure and eventually undergoes cell lysis, leading to bacterial death. Because human cells lack peptidoglycan cell walls, penicillin exhibits selective toxicity toward bacteria while causing minimal damage to host tissues.
Discovery of Sulfonamides
Another major milestone in the development of antimicrobial drugs was the discovery of sulfonamides, often referred to as “sulfa drugs.” These compounds were among the first widely used antimicrobial agents before the widespread availability of penicillin.
The antibacterial properties of sulfonamides were discovered in the early 1930s through the work of Gerhard Domagk, a German pathologist working for the pharmaceutical company Bayer. Domagk discovered that a synthetic dye known as Prontosil could successfully treat bacterial infections in experimental animals.
Further research revealed that Prontosil was metabolized in the body to produce sulfanilamide, the compound responsible for its antibacterial activity. Sulfonamides function by inhibiting the synthesis of folic acid, an essential cofactor required for bacterial growth and DNA replication.
The introduction of sulfonamides represented a major breakthrough in antimicrobial therapy and saved countless lives before the widespread use of penicillin.
The Golden Age of Antibiotic Discovery
The period between the 1940s and the 1960s is often referred to as the “Golden Age of Antibiotic Discovery.” During this time, researchers discovered and developed many of the major antibiotic classes that are still used today.
One of the key figures during this period was the Ukrainian-American microbiologist Selman Waksman, who pioneered the systematic search for antibiotic-producing microorganisms in soil. Waksman and his colleagues discovered several important antibiotics produced by soil-dwelling bacteria belonging to the genus Streptomyces.
In 1943, Waksman and his graduate student Albert Schatz discovered streptomycin, the first antibiotic effective against Mycobacterium tuberculosis, the bacterium responsible for tuberculosis. This discovery revolutionized the treatment of tuberculosis and earned Waksman the Nobel Prize in 1952.
Subsequent research during this period led to the discovery of numerous other antibiotics, including:
- Tetracyclines
- Chloramphenicol
- Aminoglycosides
- Macrolides
- Cephalosporins
- Glycopeptides
- Rifamycins
- Ciprofloxacin
Many of these antibiotics were isolated from microorganisms found in soil, marine environments, and other ecological niches.
Expansion of Antibiotic Classes
As microbiological and biochemical techniques improved, scientists began modifying naturally occurring antibiotics to improve their effectiveness, stability, and spectrum of activity. This led to the development of semi-synthetic antibiotics, which are chemically modified derivatives of natural compounds.
For example, cephalosporins were developed from a natural antibiotic produced by the fungus Cephalosporium acremonium. Through chemical modification, researchers created multiple generations of cephalosporins with improved activity against Gram-negative bacteria.
Similarly, modifications of the original penicillin molecule led to the development of several important derivatives, including:
- Methicillin
- Ampicillin
- Amoxicillin
These drugs expanded the range of bacterial infections that could be effectively treated.
Impact of Antibiotics on Public Health
The introduction of antibiotics represents one of the most significant advances in the history of modern medicine and public health. Prior to the antibiotic era, infectious diseases were among the leading causes of mortality worldwide. Common bacterial infections such as pneumonia, tuberculosis, septicemia, and wound infections frequently resulted in severe complications or death, particularly among children, the elderly, and individuals with weakened immune systems. Even minor injuries could become life-threatening if bacterial infection occurred. The discovery and widespread use of antibiotics therefore dramatically transformed clinical medicine by providing effective therapeutic tools to combat bacterial pathogens.
Antibiotics also enabled the development of modern medical procedures that would otherwise be too risky due to infection. These include:
- Major surgical operations
- Organ transplantation
- Chemotherapy for cancer
- Neonatal intensive care
Together with improvements in sanitation, vaccination programs, and access to clean water, antibiotics contributed to a substantial increase in life expectancy during the twentieth century.
One of the most immediate impacts of antibiotics was the dramatic reduction in mortality associated with infectious diseases. Antibiotics such as penicillin and later antimicrobial agents allowed physicians to effectively treat bacterial pneumonia, bloodstream infections, and postoperative infections that were previously difficult or impossible to cure. As a result, survival rates improved significantly, and hospital mortality related to bacterial infections declined in many parts of the world. The availability of reliable antimicrobial therapy also shortened recovery times and reduced complications associated with untreated infections.
Beyond treating infections directly, antibiotics have played a critical enabling role in the development and safety of modern medical procedures. Many contemporary medical interventions would carry unacceptable infection risks without effective antimicrobial agents. For instance, antibiotics are routinely used to prevent or treat infections associated with major surgical procedures, where exposure of internal tissues can allow bacterial contamination. Similarly, organ transplantation relies heavily on antibiotic prophylaxis and treatment because transplant recipients often require immunosuppressive therapies that increase their susceptibility to infection.
Antibiotics are also indispensable in cancer chemotherapy, where cytotoxic drugs suppress the immune system and make patients vulnerable to opportunistic bacterial infections. Without antibiotics, many chemotherapy regimens would pose severe infection risks. Likewise, neonatal intensive care depends on the availability of antibiotics to manage infections in premature or critically ill newborns whose immune systems are not fully developed.
In addition to their direct medical benefits, antibiotics have contributed to broader improvements in global public health. When combined with advances in sanitation, vaccination programs, improved nutrition, and access to clean water, antibiotics helped reduce the burden of infectious diseases throughout the twentieth century. These developments collectively contributed to a significant rise in global life expectancy and improved quality of life in many regions of the world. As a result, antibiotics remain a cornerstone of modern healthcare systems and continue to play an essential role in protecting public health.
Emergence of Antibiotic Resistance
Despite their remarkable benefits, the widespread use of antibiotics has led to the emergence of antibiotic-resistant bacteria. Resistance occurs when bacteria evolve mechanisms that allow them to survive exposure to antimicrobial drugs.
Alexander Fleming himself warned about this possibility in his Nobel Prize lecture in 1945, noting that misuse of penicillin could lead to the development of resistant bacteria.
Today, antimicrobial resistance is recognized as a major global health threat. Resistant pathogens such as methicillin-resistant Staphylococcus aureus (MRSA), multidrug-resistant Mycobacterium tuberculosis, and carbapenem-resistant Enterobacteriaceae (CRE) pose serious challenges to healthcare systems worldwide.
Factors contributing to antimicrobial resistance include:
- Overuse of antibiotics in human medicine
- Misuse of antibiotics in agriculture and livestock production
- Incomplete treatment courses
- Environmental contamination with antimicrobial residues
Addressing these challenges requires coordinated global efforts in surveillance, antibiotic stewardship, and the development of new antimicrobial agents.
References
Ashutosh Kar (2008). Pharmaceutical Microbiology, 1st edition. New Age International Publishers: New Delhi, India.
Bisht R., Katiyar A., Singh R and Mittal P (2009). Antibiotic Resistance – A Global Issue of Concern. Asian Journal of Pharmaceutical and Clinical Research, 2 (2):34-39.
Courvalin P, Leclercq R and Rice L.B (2010). Antibiogram. ESKA Publishing, ASM Press, Canada.
Denyer S.P., Hodges N.A and Gorman S.P (2004). Pharmaceutical Microbiology. 7th ed. Blackwell Publishing Company, USA.
Fernandes Prabhavathi (2006). Antibacterial discovery – the failure of success? Nature Biotechnology, 24(12):1.
Finch R.G, Greenwood D, Norrby R and Whitley R (2002). Antibiotic and chemotherapy, 8th edition. Churchill Livingstone, London and Edinburg.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.