Structural Characteristics and Molecular Properties
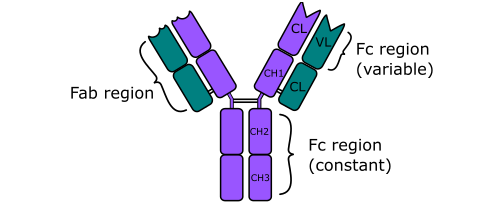
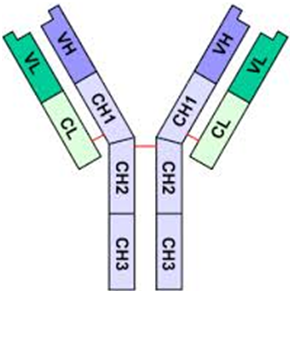
Immunoglobulin G (IgG) is a monomeric antibody that represents the most abundant class of immunoglobulins in human serum. Structurally, IgG consists of two identical heavy (γ) chains and two identical light chains linked by disulfide bonds, forming a characteristic Y-shaped configuration. Each IgG molecule contains two antigen-binding fragments (Fab regions) and one crystallizable fragment (Fc region), enabling both antigen recognition and effector function engagement (Figure 1). The molecular weight of IgG is approximately 150 kDa, and its monomeric configuration distinguishes it from other immunoglobulin classes such as IgM, which exists as a pentamer, and IgA, which commonly forms dimers in secretions. The hinge region between the Fab and Fc domains provides structural flexibility, allowing optimal antigen binding to spatially diverse epitopes.
IgG is further subdivided into four subclasses in humans: IgG1, IgG2, IgG3, and IgG4. These subclasses differ in hinge length, flexibility, and ability to activate complement or bind to Fc receptors. For example, IgG1 and IgG3 are potent activators of the classical complement pathway and exhibit strong affinity for Fcγ receptors, enhancing opsonization and antibody-dependent cellular cytotoxicity (ADCC). In contrast, IgG2 and IgG4 demonstrate reduced complement activation and distinct functional roles depending on antigen type and immune context. The structural heterogeneity among subclasses contributes to the versatility of IgG in mediating adaptive immune responses. Post-translational modifications such as glycosylation of the Fc region further modulate effector functions and influence inflammatory signaling.
Immunoglobulin G is a structurally defined 150,000 Da (150 kDa) monomeric antibody with diverse and critical immunological functions. Its moderate capacity for complement fixation contributes to pathogen opsonization and immune-mediated lysis, while its ability to mediate antibody-dependent cell-mediated cytotoxicity enables targeted elimination of infected or malignant cells. Additionally, the unique property of placental transfer grants IgG a central role in providing passive immunity to the fetus and newborn, ensuring early-life protection against infectious diseases. Collectively, these structural and functional attributes establish IgG as a pivotal component of adaptive immunity and immune homeostasis.
Role of IgG in Secondary Immune Response and Immune Memory
IgG plays a central role in the secondary or memory immune response following re-exposure to a previously encountered antigen. Upon primary antigen exposure, naïve B cells differentiate into plasma cells that initially produce IgM. With T-cell help and class-switch recombination, activated B cells subsequently switch to IgG production. During secondary exposure, memory B cells rapidly differentiate into plasma cells and produce high-affinity IgG antibodies. This response is characterized by faster kinetics, higher antibody titers, and increased affinity due to somatic hypermutation and affinity maturation in germinal centers. As a result, IgG dominates the humoral response during repeated or persistent antigen challenges. IgG mediates pathogen neutralization by binding to viral particles and toxins, thereby preventing their interaction with host cell receptors. It also facilitates opsonization, where IgG-coated pathogens are recognized by Fcγ receptors on phagocytic cells such as macrophages and neutrophils. This interaction enhances phagocytosis and intracellular destruction of pathogens.
Additionally, IgG activates the classical complement pathway through binding of its Fc region to complement component C1q. Complement activation leads to pathogen lysis via the membrane attack complex and amplifies inflammatory signaling to recruit immune effector cells. These mechanisms collectively make IgG a critical mediator of protective immunity against bacterial, viral, and some parasitic infections. The persistence of IgG in circulation contributes to long-term immune surveillance. Its relatively long half-life approximately 21 days in humans is regulated by the neonatal Fc receptor (FcRn), which rescues IgG from lysosomal degradation and recycles it back into circulation. This recycling mechanism enhances serum stability and maintains sustained immune protection.
Placental Transfer of IgG and its Protective Immunity in Neonates
One of the most distinctive biological properties of IgG is its ability to cross the placental barrier. Unlike other immunoglobulin classes, IgG is actively transported from the maternal circulation to the fetal bloodstream through FcRn-mediated transcytosis in placental syncytiotrophoblasts. This selective transfer begins in the second trimester of pregnancy and increases significantly during the third trimester, resulting in higher concentrations of maternal IgG in the newborn at birth. Consequently, neonates acquire passive humoral immunity that protects them against pathogens encountered by the mother. Maternal IgG provides temporary immune defense during the first six months of life, a period when the infant’s immune system is still immature and has limited capacity to mount robust antibody responses. This passive immunity is particularly important for protection against respiratory infections, systemic bacterial infections, and certain viral diseases.
The effectiveness of placental IgG transfer depends on subclass specificity and maternal antibody levels. IgG1 is transferred most efficiently, followed by IgG4, IgG3, and IgG2. Vaccination during pregnancy leverages this physiological mechanism to enhance neonatal immunity against preventable infectious diseases such as influenza, pertussis, and tetanus. However, maternal IgG can also have clinical implications beyond protection. In some cases, pathogenic autoantibodies or alloantibodies cross the placenta and contribute to neonatal autoimmune or hemolytic conditions. This demonstrates that while placental IgG transfer is protective, it can also mediate disease under certain immunological circumstances.
Immunoglobulin G is the most predominant antibody class in blood serum and extracellular fluids, accounting for approximately 80% of the total immunoglobulin pool. Its monomeric structure, subclass diversity, and functional versatility enable it to play a central role in adaptive immunity. Through participation in secondary immune responses, IgG ensures rapid and high-affinity antibody production upon antigen re-exposure. Its ability to neutralize pathogens, mediate opsonization, activate complement, and engage Fc receptors underscores its importance in immune defense. Importantly, IgG is the only immunoglobulin class capable of crossing the placental barrier, thereby conferring passive immunity to the newborn during the early months of life. This transplacental transfer highlights its critical role in intergenerational immune protection. IgG represents a fundamental component of humoral immunity, bridging innate immune mechanisms with long-term adaptive protection and contributing to both clinical immunity and vaccine-mediated disease prevention.
Role of IgG in Passive Immunity and Maternal Fetal Protection
A distinctive biological feature of IgG is its ability to cross the placenta, providing passive immunity from mother to fetus. This transplacental transfer is mediated by the neonatal Fc receptor (FcRn), which actively transports IgG across placental barrier cells. Among immunoglobulin classes, IgG is the only class that efficiently crosses the placenta in significant amounts. Placental transfer of IgG typically increases during the second and third trimesters of pregnancy, leading to accumulation of maternal antibodies in the fetal circulation. This mechanism ensures that neonates are born with a repertoire of protective antibodies against pathogens to which the mother has previously been exposed or vaccinated against. Consequently, newborns acquire naturally transferred passive immunity that provides early protection during a period when their own adaptive immune system is still immature.
After birth, maternally derived IgG continues to offer protection through circulation in the infant’s bloodstream. Although antibody levels gradually decline over months as maternal IgG is catabolized, this temporal protection bridges the gap until the infant develops robust endogenous antibody production. This biological function is particularly important for protection against respiratory pathogens, bacterial infections, and certain viral diseases during early life. Beyond fetal protection, passive immunization strategies in clinical medicine often utilize purified IgG preparations to confer temporary immunity in individuals with immunodeficiency or acute exposure to specific pathogens. Intravenous immunoglobulin (IVIG) therapy exemplifies the therapeutic application of pooled IgG to modulate immune responses and enhance immune defense.
Complement Activation and Antibody-Dependent Cellular Cytotoxicity
One of the key immunological functions of IgG is its ability to activate the complement system, although it is considered a moderate complement-fixing antibody relative to other immunoglobulin classes. Complement activation primarily occurs through the classical pathway, which is initiated when the Fc region of antigen-bound IgG interacts with the C1 complex. This interaction triggers a cascade of proteolytic events leading to opsonization, inflammation, and the formation of the membrane attack complex. The efficiency of complement activation depends on antibody density, subclass type, and spatial arrangement of IgG molecules on antigen surfaces. IgG3 and IgG1 subclasses demonstrate stronger complement fixation because of their Fc structural configuration, whereas IgG4 exhibits minimal complement activation capacity. Through complement deposition, pathogens are marked for enhanced phagocytosis by macrophages and neutrophils, contributing to pathogen clearance.
In addition to complement activation, IgG mediates antibody-dependent cell-mediated cytotoxicity (ADCC), an essential effector mechanism in immune defense against infected or transformed cells. ADCC occurs when IgG antibodies bind to specific antigens expressed on target cells. Effector cells such as natural killer (NK) cells, macrophages, and neutrophils express Fc gamma receptors (FcγRs) that recognize the Fc portion of bound IgG. Upon FcγR engagement, effector cells release cytotoxic molecules including perforin and granzymes or produce inflammatory mediators that induce apoptosis of the target cell. This mechanism is particularly important in viral infections, tumor surveillance, and clearance of antibody-coated cells. The effectiveness of ADCC depends on the affinity between IgG Fc domains and Fc receptors, as well as the level of antigen expression on target cells. Therapeutic monoclonal antibodies used in oncology frequently exploit ADCC as a major mechanism of action.
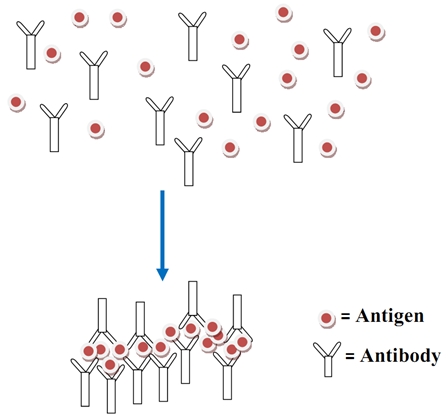
Immunoglobulin G (IgG) is typically produced during the later stages of an immune response and remains detectable in the circulation for an extended period compared with other immunoglobulin isotypes. Its delayed appearance reflects class-switch recombination and affinity maturation processes that occur following initial antigen exposure. Once generated, IgG contributes to long-term humoral immunity and serves as a key marker of past or ongoing infection. IgG binds specifically to antigens expressed on pathogenic microorganisms, including bacteria and viruses. This antigen–antibody interaction promotes immune clearance through several effector mechanisms. One of the primary functions of IgG is opsonization, whereby the antibody coats the surface of the pathogen and enhances its recognition by phagocytic cells such as macrophages and neutrophils. The Fc region of IgG interacts with Fc gamma receptors on phagocytes, facilitating efficient engulfment and intracellular destruction of the targeted microorganism. As a result, opsonized microbes are phagocytosed more readily than non-opsonized antigens, significantly improving host defense. Beyond opsonization, IgG also participates in complement activation via the classical pathway, leading to the deposition of complement components on microbial surfaces. This process further enhances phagocytosis and can directly contribute to microbial lysis. In addition, IgG mediates antibody-dependent cellular cytotoxicity (ADCC), where immune effector cells recognize IgG-coated targets and induce targeted cell death. These combined mechanisms underscore the central role of IgG in adaptive immunity and pathogen clearance.
IgG is widely utilized in immunological research and diagnostic applications because of its relatively high concentration in serum and plasma, as well as its strong affinity and specificity for antigens. Its stability and well-characterized structure make it suitable for assays such as enzyme-linked immunosorbent assays (ELISA), immunoblotting, immunofluorescence, and neutralization studies. Furthermore, purified IgG antibodies are commonly employed as research reagents and therapeutic agents due to their predictable binding properties and functional versatility.
Role of IgG in Immunological Research and Diagnostic Applications
Immunoglobulin G (IgG) plays a central role in immunology as both a functional effector molecule in adaptive immunity and a critical tool in diagnostic and translational research. Its biochemical stability, high specificity, and abundance in serum make it indispensable for experimental immunology, disease diagnostics, vaccine evaluation, and therapeutic development. IgG continues to remains a cornerstone of immunological experimentation, diagnostic assay development, and therapeutic innovation due to its specificity, functional versatility, and clinical relevance.
In immunological research, IgG is widely used as a detection and analytical reagent. Purified IgG antibodies are generated against specific antigens and employed in techniques such as enzyme-linked immunosorbent assay (ELISA), Western blotting, immunoprecipitation, immunofluorescence microscopy, and flow cytometry. In these applications, IgG serves as a primary antibody that recognizes target proteins, pathogens, or cellular markers with high specificity. Secondary antibodies often anti-IgG conjugated to enzymes, fluorophores, or nanoparticles are used to amplify detection signals. This dual-antibody system enhances assay sensitivity and allows quantitative or qualitative measurement of antigens.
IgG is also fundamental in studying immune responses to infection and vaccination. The measurement of antigen-specific IgG titers provides insight into humoral immunity and immune memory. In vaccine research, quantification of vaccine-induced IgG responses is used as a correlate of protection, helping researchers assess immunogenicity and durability of immune responses. Neutralizing IgG antibodies, which block pathogen entry into host cells, are particularly important in evaluating vaccine efficacy against viral infections. Longitudinal monitoring of IgG levels enables assessment of antibody kinetics, seroconversion rates, and long-term immune persistence.
In diagnostic applications, IgG-based assays are extensively used for serological testing. Detection of pathogen-specific IgG in patient serum indicates prior exposure or infection. For many infectious diseases, including viral, bacterial, and parasitic infections, IgG testing is used to determine immune status, past infection, or vaccine-induced immunity. Because IgG persists longer in circulation compared with IgM or IgA, it serves as a reliable biomarker for historical exposure. Diagnostic platforms such as rapid diagnostic tests, lateral flow immunoassays, and chemiluminescent immunoassays commonly rely on IgG detection for clinical screening and surveillance.
Beyond infectious disease diagnostics, IgG is important in autoimmune disease research and clinical testing. Autoantibodies (which are self-reactive IgG molecules) are key biomarkers in conditions such as systemic lupus erythematosus, rheumatoid arthritis, and autoimmune thyroid disease. Detection of disease-associated IgG autoantibodies assists in diagnosis, disease classification, and monitoring of disease progression. In addition, characterization of IgG subclasses helps researchers understand pathogenic mechanisms, as different subclasses exhibit distinct effector functions and inflammatory potentials.
Therapeutically, IgG forms the basis of monoclonal antibody (mAb) development and antibody-based biologics. Engineered IgG antibodies are used to target cancer cells, neutralize toxins, inhibit inflammatory pathways, or block viral entry. Advances in recombinant DNA technology and hybridoma techniques have enabled the production of highly specific monoclonal IgG antibodies for clinical use. Intravenous immunoglobulin (IVIG), which consists primarily of pooled IgG from donors, is also applied in immunodeficiency treatment and autoimmune disease management due to its immunomodulatory properties.
IgG is utilized in biomarker discovery and proteomics research. Antibody-based affinity purification enables enrichment of specific proteins or immune complexes from biological samples for downstream mass spectrometry analysis. This approach facilitates identification of disease-associated proteins and signaling molecules.
REFERENCES
Abbas A.K, Lichtman A.H and Pillai S (2010). Cellular and Molecular Immunology. Sixth edition. Saunders Elsevier Inc, USA.
Actor J (2014). Introductory Immunology. First edition. Academic Press, USA.
Alberts B, Bray D, Johnson A, Lewis J, Raff M, Roberts K and Walter P (1998). Essential Cell Biology: An Introduction to the Molecular Biology of the Cell. Third edition. Garland Publishing Inc., New York.
Bach F and Sachs D (1987). Transplantation immunology. N. Engl. J. Med. 317(8):402-409.
Barrett J.T (1998). Microbiology and Immunology Concepts. Philadelphia, PA: Lippincott-Raven Publishers. USA.
Jaypal V (2007). Fundamentals of Medical Immunology. First edition. Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India.
John T.J and Samuel R (2000). Herd Immunity and Herd Effect: New Insights and Definitions. European Journal of Epidemiology, 16:601-606.
Levinson W (2010). Review of Medical Microbiology and Immunology. Twelfth edition. The McGraw-Hill Companies, USA.
Roitt I, Brostoff J and Male D (2001). Immunology. Sixth edition. Harcourt Publishers Limited, Spain.
Zon LI (1995). Developmental biology of hematopoiesis. Blood, 86(8): 2876–91.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.