People are often confused by differences between Good Laboratory Practice (GLP) regulations and Good Manufacturing Practice (GMP) […]
Tag: GMP
RULES OF GOOD MANUFACTURING PRACTICE (GMP)
GMP encompasses the rules governing the manufacture of a safe and efficacious pharmaceutical product, drug […]
CRITICAL ASPECTS OF GMP (sources of microbial contamination)
Some of the main sources of contamination in the course of production in a food […]
PRINCIPLES OF GOOD MANUFACTURING PRACTICE (GMP)
GMP guidelines are not prescriptive instructions on how to manufacture any product including food, drugs, […]
GOOD MANUFACTURING PRACTICE (GMP)
Good manufacturing practice (GMP) is simply defined as those general rules that govern the manufacture […]
Quality Assurance: System Design and Implementation in Pharmaceutical Manufacturing and Clinical Laboratory Practice
Quality Assurance (QA) is a structured, systematic, and documented framework designed to ensure that products […]
Quality Control in Pharmaceutical Manufacturing: Principles, Microbial Testing, Practical Implementation & GMP Compliance
Introduction Quality control (QC) is a systematic monitoring and verification framework used to detect, measure, […]
PYROGEN TEST
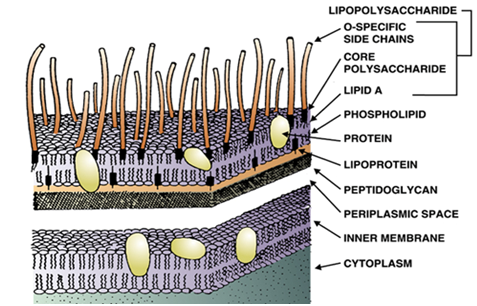
A pyrogen is simply defined as a fever-causing (inducing) agent that includes toxins of microorganisms. […]