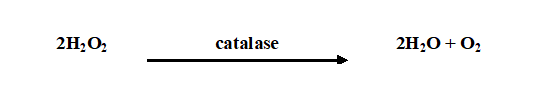
Methyl red test is used to identify pathogenic bacteria that produce acid from glucose phosphate. Some bacterial isolates ferment glucose through the mixed acid fermentation pathway to produce several acids including acetic acid, formic acid and lactic acid.
Production of steady amounts of acids by these microorganisms will result in a pH level that is usually below 4.4 (acidic) because the phosphate buffer of the medium have been overwhelmed by the acid produced.
Addition of methyl red to the medium result in this acidic medium and a red colouration is produced. Escherichia coli and Proteus species are both methyl red positive.
PROCEDURE FOR METHYL RED TEST
- Prepare and dispense glucose phosphate broth in 9 ml portions of clean test tubes and sterilize the medium by autoclaving.
- Bring the prepared medium to room temperature and allow to cool after sterilization.
- Inoculate the test isolate in duplicates.
- Dissolve 0.1 g of methyl red powder in 300 ml of 95 % ethanol.
- Make up the solution to 500 ml using distilled water.
- Add few drops of the prepared methyl red solution (about 3-5 drops) to the broth culture of the test bacteria while leaving one of the tubes uninoculated with methyl red. This tube will serve as the negative control tube. A red colour appears in the tube(s) when the pH indicator (methyl red) is added to the broth culture, and this is possible if the pH of the broth culture medium is actually below 4.4.
- Observe the tube(s) for a red colour which shows a positive methyl red test result. A yellow colouration shows a negative result (Figure 1).

References
Basic laboratory procedures in clinical bacteriology. World Health Organization (WHO), 1991. Available from WHO publications, 1211 Geneva, 27-Switzerland.
Beers M.H., Porter R.S., Jones T.V., Kaplan J.L and Berkwits M (2006). The Merck Manual of Diagnosis and Therapy. Eighteenth edition. Merck & Co., Inc, USA.
Biosafety in Microbiological and Biomedical Laboratories. 5th edition. U.S Department of Health and Human Services. Public Health Service. Center for Disease Control and Prevention. National Institute of Health. HHS Publication No. (CDC) 21-1112.2009.
Cheesbrough M (2010). District Laboratory Practice in Tropical Countries. Part I. 2nd edition. Cambridge University Press, UK.
Cheesbrough M (2010). District Laboratory Practice in Tropical Countries. Part 2. 2nd edition. Cambridge University Press, UK.
Collins C.H, Lyne P.M, Grange J.M and Falkinham J.O (2004). Collins and Lyne’s Microbiological Methods. Eight edition. Arnold publishers, New York, USA.
Disinfection and Sterilization. (1993). Laboratory Biosafety Manual (2nd ed., pp. 60-70). Geneva: WHO.
Garcia L.S (2010). Clinical Microbiology Procedures Handbook. Third edition. American Society of Microbiology Press, USA.
Garcia L.S (2014). Clinical Laboratory Management. First edition. American Society of Microbiology Press, USA.
Fleming, D. O., Richardson, J. H., Tulis, J. I. and Vesley, D. (eds) (1995). Laboratory Safety: Principles and practice. Washington DC: ASM press.
Dubey, R. C. and Maheshwari, D. K. (2004). Practical Microbiology. S.Chand and Company LTD, New Delhi, India.
Gillespie S.H and Bamford K.B (2012). Medical Microbiology and Infection at a glance. 4th edition. Wiley-Blackwell Publishers, UK.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.