Innate immunity constitutes the host’s intrinsic, evolutionarily conserved defense system and represents the first immunological barrier against invading microorganisms. It is present at birth and operates independently of prior antigenic exposure. Unlike adaptive immunity, which requires clonal expansion and antigen-specific receptor rearrangement, innate immunity provides an immediate response to infection through preformed cellular and molecular mechanisms. This rapid reactivity is critical during the early stages of pathogen invasion, when microbial replication may otherwise outpace the development of adaptive immune responses.
Fundamentally, innate immunity is nonspecific in its mode of action. It does not target a particular pathogen or antigenic epitope; rather, it recognizes conserved molecular structures that are broadly shared among classes of microorganisms. These conserved motifs, often referred to as pathogen-associated molecular patterns (PAMPs), include lipopolysaccharide in Gram-negative bacteria, peptidoglycan in Gram-positive bacteria, flagellin, viral double-stranded RNA, and unmethylated cytosine-phosphate-guanine (CpG) DNA. Host cells detect these structures through germline-encoded pattern recognition receptors (PRRs), such as Toll-like receptors (TLRs), NOD-like receptors, and RIG-I-like receptors. Because these receptors are genetically predetermined and not somatically rearranged, their specificity does not change over the lifespan of the individual.
The components of innate immunity can be broadly categorized into physical barriers, cellular defenses, and soluble mediators. Physical and anatomical barriers constitute the first protective layer. The intact skin, with its keratinized epithelium and acidic surface pH, prevents microbial entry, while mucosal surfaces are protected by mucus, ciliary action, and antimicrobial peptides such as defensins and lysozyme. These barriers reduce the probability of pathogen penetration and colonization.
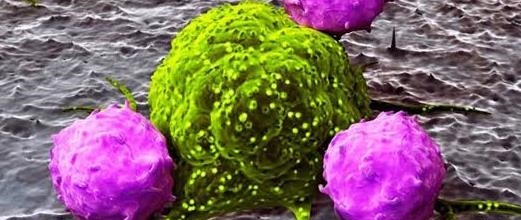
If pathogens breach these barriers, cellular components of innate immunity are rapidly mobilized. Key effector cells include neutrophils, macrophages, dendritic cells, and natural killer (NK) cells. Neutrophils are typically the first leukocytes recruited to sites of infection, where they phagocytose and destroy microbes through oxidative burst and enzymatic degradation. Macrophages perform similar phagocytic functions but also act as antigen-presenting cells and produce pro-inflammatory cytokines that orchestrate the inflammatory response. Dendritic cells serve as a critical interface between innate and adaptive immunity by capturing antigens and migrating to lymphoid tissues to activate T lymphocytes. NK cells contribute to early antiviral defense by recognizing and eliminating infected or transformed cells through cytotoxic mechanisms independent of prior sensitization.
Soluble factors further amplify innate immune responses. The complement system, a cascade of plasma proteins, enhances opsonization, promotes inflammation, and directly lyses susceptible pathogens. Acute-phase proteins, cytokines (such as interleukins and tumor necrosis factor), and chemokines coordinate leukocyte recruitment and systemic responses, including fever and inflammation. Collectively, these mechanisms limit pathogen dissemination and create conditions conducive to adaptive immune activation.
A defining characteristic of innate immunity is its lack of immunological memory. The magnitude and quality of the innate response remain essentially constant upon repeated exposure to the same pathogen. Unlike adaptive immunity, which improves in speed and specificity following re-exposure due to memory B and T cells, innate responses are considered static because their receptor repertoire is fixed and non-clonal. Although emerging evidence suggests phenomena such as “trained immunity” in certain innate cells, this does not equate to the highly specific, long-lasting memory characteristic of adaptive immune responses.
Innate immunity is a rapid, nonspecific, and genetically predetermined defense system that provides immediate protection against a wide array of pathogens. By functioning as the first line of defense and by shaping subsequent adaptive responses, it plays an indispensable role in maintaining host integrity and preventing infection.
Innate immunity constitutes the host’s first line of defense against invading microorganisms. It is evolutionarily conserved, rapid in onset, and non–antigen specific, functioning independently of prior exposure to a pathogen. Unlike adaptive immunity, which requires clonal expansion and immunologic memory, innate immune mechanisms respond immediately to conserved microbial structures known as pathogen-associated molecular patterns (PAMPs). Innate immunity encompasses integrated physical, chemical, cellular, and microbial defenses that operate synergistically to prevent infection. These mechanisms act rapidly and broadly against diverse pathogens, providing essential protection while adaptive immune responses are mobilized.
The principal components of innate immunity can be organized into five major categories of defensive barriers:
- Anatomic barriers,
- Physiologic barriers,
- Phagocytic barriers,
- Inflammatory barriers, and
- The intact normal microflora (microbiota) – which forms a critical protective interface that complements these host-derived mechanisms.
1. Anatomic (Physical and Mechanical) Barriers
Anatomic barriers provide the first physical impediment to microbial entry. The skin is the most extensive of these barriers. Its outermost layer, the stratum corneum, consists of keratinized epithelial cells embedded in a lipid matrix, creating a relatively impermeable surface. Continuous desquamation further limits microbial colonization by mechanically removing adherent organisms.
Mucosal surfaces lining the respiratory, gastrointestinal, and genitourinary tracts represent additional anatomic barriers. Although structurally thinner than skin, these epithelia are reinforced by mucus secretions that trap microorganisms. In the respiratory tract, ciliated epithelial cells facilitate mucociliary clearance, propelling trapped particles and microbes upward toward the pharynx for elimination. In the gastrointestinal tract (GIT), peristalsis and the continuous flow of luminal contents reduce microbial adherence and overgrowth. Similarly, the flushing action of urine in the urinogenital tract mechanically removes potential pathogens.
Other specialized epithelial surfaces, including the mammary gland epithelium and conjunctival membranes, contribute localized protective functions. Tight junctions between epithelial cells limit paracellular penetration of microbes, while epithelial cells themselves produce antimicrobial substances that reinforce barrier integrity.
2. Physiologic and Biochemical Barriers
Physiologic barriers create inhospitable conditions that restrict microbial survival and replication. Body temperature, particularly during fever, inhibits the growth of temperature-sensitive pathogens and enhances certain immune functions. Fever is mediated by endogenous pyrogens such as interleukin-1 (IL-1) and tumor necrosis factor (TNF), which act on the hypothalamus to elevate the thermal set point.
pH is another critical determinant. The acidic environment of the stomach (pH 1.5–3.5) destroys many ingested microorganisms. Likewise, the mildly acidic pH of the skin and vagina limits colonization by pathogenic species.
A wide array of chemical mediators further strengthens innate defenses. Lysozyme, present in tears, saliva, and other secretions, hydrolyzes peptidoglycan in bacterial cell walls, particularly in Gram-positive organisms. Antimicrobial peptides (e.g., defensins and cathelicidins) disrupt microbial membranes. Surfactant proteins in the lungs enhance opsonization and microbial clearance. Lipids and fatty acids on the skin possess bactericidal properties.
Cytokines and interleukins orchestrate early immune signaling, while interferons (especially type I interferons) inhibit viral replication within infected and neighboring cells. The complement system, a cascade of serum proteins, promotes opsonization, inflammation, and direct microbial lysis through the formation of the membrane attack complex (MAC). Collectively, these physiologic and biochemical mechanisms create a hostile microenvironment for invading pathogens.
3. Phagocytic Barriers
Phagocytic cells represent a central cellular component of innate immunity. These specialized leukocytes identify, engulf, and destroy invading microorganisms through a process known as phagocytosis.
Neutrophils are typically the first responders to sites of infection. They migrate via chemotaxis in response to inflammatory mediators and microbial products. Once at the site, they internalize pathogens into phagosomes, which fuse with lysosomes to form phagolysosomes where reactive oxygen species (ROS), proteolytic enzymes, and antimicrobial peptides mediate intracellular killing.
Monocytes circulate in the bloodstream and differentiate into macrophages upon entering tissues. Macrophages perform sustained phagocytic activity, produce pro-inflammatory cytokines, and serve as antigen-presenting cells bridging innate and adaptive immunity. Eosinophils, although less prominent in bacterial infections, contribute particularly to defense against helminths and in allergic responses by releasing cytotoxic granule proteins.
Phagocytosis is enhanced by opsonization, whereby complement proteins (e.g., C3b) or antibodies coat microbial surfaces, facilitating recognition by phagocyte receptors. Through these mechanisms, phagocytic cells not only eliminate pathogens but also regulate the magnitude and duration of inflammatory responses.
4. Inflammatory Barrier
Inflammation is a coordinated vascular and cellular response triggered by tissue injury or infection. It serves to contain and eliminate pathogens while initiating tissue repair. The classical signs of inflammation – redness (rubor), heat (calor), swelling (tumor), and pain (dolor) – reflect underlying physiologic changes.
Vasodilation increases blood flow to the affected area, accounting for redness and heat. Enhanced vascular permeability permits plasma proteins, including complement components and clotting factors, to exit the circulation and enter the interstitial space. This exudation results in edema (swelling). Pain arises from the stimulation of sensory nerve endings by inflammatory mediators such as prostaglandins and bradykinin.
Simultaneously, leukocytes adhere to activated endothelium and migrate into tissues via diapedesis. The accumulation of phagocytic cells and serum proteins at the infection site restricts microbial spread and facilitates pathogen clearance. Although inflammation is protective, excessive or dysregulated inflammation can contribute to tissue damage.
5. Intact Normal Microflora (Microbiota)
The normal microflora comprising commensal bacteria and fungi inhabiting the skin and mucosal surfaces constitute an additional, often underappreciated, component of innate defense. These microbial communities may be resident (permanent colonizers) or transient.
The microbiota confer protection primarily through competitive exclusion. By occupying ecological niches and competing for nutrients and attachment sites, they limit colonization by pathogenic organisms. Many commensals also produce bacteriocins and metabolic byproducts that inhibit pathogen growth. In the gastrointestinal tract, certain microbial species contribute to immune maturation and modulate inflammatory responses. Disruption of normal microflora, such as through broad-spectrum antibiotic use, can predispose individuals to opportunistic infections by diminishing this protective barrier.
Normal Microbiota as a Primary Mechanism of Microbial Antagonism in the Human Body
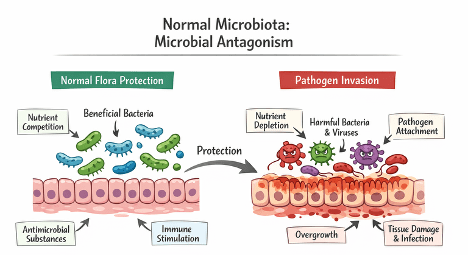
The normal microbiota constitutes one of the most important natural defenses against pathogenic invasion through a process known as microbial antagonism. The human body exists in continuous association with a diverse consortium of microorganisms, including bacteria, fungi, and viruses, that colonize surfaces such as the skin, gastrointestinal tract, respiratory tract, and genitourinary system. These resident microbial communities form a dynamic ecological barrier that limits the establishment and proliferation of pathogenic organisms.
Microbial antagonism occurs when the indigenous microbiota inhibits the colonization and growth of exogenous or opportunistic pathogens. This protection is mediated through several well-characterized mechanisms. First, normal flora competes with invading microbes for ecological niches and adhesion sites on epithelial surfaces, thereby preventing pathogen attachment – a critical initial step in infection. Second, they compete for essential nutrients such as carbon sources, amino acids, iron, and vitamins, depriving potential pathogens of the metabolic resources required for replication.
Microbial antagonism refers to the protective interaction in which established normal microbiota or beneficial microorganisms inhibit the colonization and growth of pathogenic organisms. This inhibition occurs primarily through competition for essential resources such as nutrients and ecological niches, thereby limiting pathogen attachment and proliferation. In addition, resident microbes produce inhibitory compounds—including bacteriocins, antimicrobial toxins, organic acids, and other metabolic byproducts – that suppress or eliminate competing pathogens. Together, these mechanisms maintain microbial balance and contribute to host defense against infection. This natural defense mechanism is vital for maintaining health, preventing infection, and controlling microbial communities.
Key Mechanisms of Microbial Antagonism
Competition for Resources: Normal microbiota compete with invading pathogens for nutrients and physical space on host tissues (Figure 1). By occupying available niches and consuming essential metabolic substrates, they limit the ability of pathogens to adhere, establish, and proliferate.
Production of Inhibitory Substances (Antibiosis): Resident microbes secrete metabolic byproducts that suppress or kill competing organisms. Examples include organic acids such as lactic acid, which lower environmental pH, and bacteriocins, which disrupt the cell membranes of susceptible microbes.
Contact-Dependent Killing: Certain bacteria utilize specialized secretion systems, such as the Type VI Secretion System (T6SS), to directly deliver toxic effector molecules into neighboring cells, leading to growth inhibition or cell death.
Environmental Modification: Microorganisms alter local conditions to create unfavorable environments for pathogens. This includes modifying pH, depleting oxygen or nutrients, and secreting enzymes such as chitinases that degrade structural components of competing organisms.
Examples and Applications of Microbial Antagonism
Human Health: Vaginal microbiota produce lactic acid that inhibits the growth of Candida albicans, while skin-associated bacteria generate fatty acids and antimicrobial compounds that protect against pathogenic colonization of the female vagina.
Agriculture: Beneficial microbes such as Trichoderma species and Bacillus spp. are applied in compost and soil to suppress soilborne plant pathogens through competitive exclusion and antagonistic interactions.
Food Safety: Lactic acid bacteria (LAB) are widely used as natural biopreservatives in food systems. They inhibit spoilage organisms and foodborne pathogens through acidification and antimicrobial compound production.
In addition to competitive exclusion, normal microbiota produces antimicrobial substances, including bacteriocins, organic acids, hydrogen peroxide, and short-chain fatty acids. These metabolites create unfavorable microenvironments – for example, by lowering pH – that suppress pathogen viability. Furthermore, the microbiota plays an essential role in modulating host immune responses. They stimulate the development and maintenance of both innate and adaptive immunity, enhancing mucosal barrier function and promoting immune surveillance.
When the normal microbial balance is disrupted – a condition termed dysbiosis – this antagonistic protection is compromised. Factors such as broad-spectrum antibiotic use, immunosuppression, poor nutrition, or disease can reduce microbial diversity and density, allowing opportunistic pathogens to colonize, proliferate, and cause infection. Thus, the normal microbiota function not merely as passive commensals but as active participants in host defense, forming a critical biological barrier against disease.
Host Determinants and Modulators of Innate Immune Response
In healthy individuals, most microorganisms encountered by the human body are rapidly recognized, contained, and eliminated by mechanisms of the innate (nonspecific) immune system before the activation of adaptive immune responses against foreign antigens. The innate immune system serves as the primary and immediate line of defense against invading pathogens. It acts at the initial stage of exposure, providing rapid protective mechanisms through physical barriers, phagocytic cells, complement proteins, and inflammatory mediators. Because the adaptive (specific) immune response requires time for antigen recognition, clonal expansion, and differentiation of effector cells, the innate response is critical in controlling infection during the early phase of host–pathogen interaction.
Innate immunity relies on germline-encoded pattern recognition receptors (PRRs) that detect conserved molecular structures on pathogens, enabling immediate immune activation. Cells such as macrophages, neutrophils, dendritic cells, and natural killer cells play central roles in pathogen recognition and clearance. In addition, soluble factors including cytokines, chemokines, and complement components amplify immune signaling and facilitate recruitment of immune cells to sites of infection. Although these mechanisms function efficiently in most individuals, their effectiveness can vary considerably depending on host-related factors.
Several intrinsic host factors influence the magnitude and quality of innate immune responses. Age is a major determinant. Newborns and infants possess an immature immune system characterized by reduced phagocytic activity, limited cytokine production, and incomplete development of immune cell function. While maternal antibodies provide partial protection, the underdeveloped innate and adaptive immune components increase susceptibility to infections. In contrast, older adults experience immunosenescence, a progressive decline in immune competence associated with reduced production of immune cells, impaired signaling pathways, and diminished responsiveness to pathogens and vaccines. These age-related alterations significantly affect infection outcomes.
Sex is another factor contributing to variability in immune responses. Immunological differences between males and females are influenced by genetic, hormonal, and epigenetic mechanisms. Certain infectious and autoimmune diseases show sex-specific prevalence or severity, partly due to differences in sex hormones such as estrogen, progesterone, and testosterone, which modulate immune cell activity and cytokine production. As a result, biological sex can shape susceptibility to infection and the intensity of inflammatory responses.
Nutritional status also plays a fundamental role in regulating innate immunity. Malnutrition, including deficiencies in essential vitamins, minerals, and macronutrients, weakens immune function and compromises the body’s ability to mount effective defense mechanisms. Undernourishment impairs barrier integrity, reduces the activity of phagocytic cells, and disrupts cytokine signaling, thereby increasing vulnerability to opportunistic and pathogenic infections. Conversely, balanced nutrition supports immune cell development, antibody production, and optimal inflammatory regulation.
Host genetic determinants further contribute to inter-individual variability in immune responses. Genetic polymorphisms in immune-related genes can influence the expression and function of pattern recognition receptors, cytokines, and signaling molecules. Such variations may enhance resistance to certain pathogens or predispose individuals to heightened inflammatory reactions and disease susceptibility. Therefore, genetic background partially explains differences in infection outcomes among individuals exposed to similar environmental conditions.
Physiological stress, fatigue, and hormonal imbalance also modulate immune function. Chronic stress triggers the prolonged release of stress hormones such as cortisol, which can suppress immune cell activity and reduce inflammatory responses. Persistent fatigue and disrupted circadian rhythms similarly impair immune regulation. Hormonal fluctuations, particularly those affecting endocrine pathways, may alter immune cell communication and responsiveness, leading to changes in host defense capacity.
While the innate immune system provides rapid and essential protection against microbial invasion, its effectiveness is influenced by multiple host-specific determinants. Age, sex, nutritional status, genetic background, stress levels, and hormonal activity collectively shape individual variations in immune competence and susceptibility to infection. Understanding these modulatory factors is essential for explaining differences in disease outcomes and for developing targeted preventive and therapeutic strategies.
Mechanisms of Innate Immunity
Innate immunity comprises rapid, non-specific defense mechanisms that protect the body against microbial invasion. Physical and chemical barriers such as the skin and mucous membranes prevent pathogen entry through structural integrity, acidic environments, and antimicrobial secretions. Phagocytic cells ingest and destroy microbes, while cytokines coordinate and regulate inflammatory and immune responses. The complement system enhances microbial killing through opsonization, inflammation, and membrane attack complex formation. Additional protective mechanisms include antimicrobial components in mammary gland secretions and colostrum, which provide passive immunity. Together, these interconnected systems act rapidly to contain infection and maintain host homeostasis.
The Skin
The skin constitutes the primary physical and chemical barrier of the body against microbial invasion. It is a continuous, keratinized epithelial surface that is structurally dense, tough, and relatively impermeable to most pathogens. As long as its integrity remains intact, it effectively blocks the entry of bacteria, viruses, fungi, and parasites. However, when the skin barrier is disrupted through burns, surgical incisions, needle pricks, insect bites, abrasions, or traumatic wounds, microorganisms gain direct access to underlying tissues and the bloodstream, increasing the risk of infection.
Beyond its structural role, the skin provides important biochemical defenses. It maintains a slightly acidic surface environment with a pH typically ranging between 3 and 5. This acidic condition inhibits the growth and survival of many pathogenic microorganisms that prefer neutral pH conditions. Sweat and sebaceous gland secretions contribute to this protective milieu by containing antimicrobial substances such as fatty acids, lactic acid, and other lipid-derived compounds that suppress microbial proliferation.
The skin also produces antimicrobial peptides and enzymes that enhance its defensive capacity. Lysozyme, an enzyme capable of degrading the peptidoglycan layer of bacterial cell walls, is present in skin secretions and contributes to the destruction of susceptible bacteria. In addition, the dry nature of the skin limits microbial colonization because most microorganisms require moisture for optimal growth. The resident commensal microbiota further strengthens protection through competitive exclusion, occupying ecological niches and limiting space and nutrients available for pathogenic organisms.
Collectively, the mechanical barrier function, acidic environment, antimicrobial secretions, and resident microbial community act synergistically to prevent pathogen establishment and invasion.
Mucous Membranes
Mucous membranes line internal body surfaces that are exposed to the external environment, including the nasal passages, oral cavity, respiratory tract, gastrointestinal tract, urinogenital tract, and cervicovaginal tract. These surfaces represent common portals of microbial entry. Their primary defensive role is to trap and eliminate invading microorganisms before they penetrate deeper tissues.
Mucus is a viscous biological secretion produced by epithelial goblet cells and submucosal glands. It forms a protective gel-like layer over epithelial surfaces, physically trapping inhaled, ingested, or introduced pathogens. Mucus contains antimicrobial components such as lysozyme, defensins, lactoferrin, and other bactericidal or bacteriostatic molecules that directly inhibit microbial growth. Lysozyme functions by hydrolyzing peptidoglycan in bacterial cell walls, leading to bacterial lysis.
A critical early step in infection is microbial adhesion to epithelial cells. Mucous secretions interfere with this process by preventing direct contact between pathogens and epithelial receptors. Secretory immunoglobulin A (sIgA), present in mucosal secretions, binds to microorganisms and neutralizes them, thereby blocking attachment and subsequent invasion. This immune exclusion mechanism significantly reduces the likelihood of colonization.
Microbes trapped within mucus are removed through coordinated mechanical processes. In the respiratory tract, ciliary movements propel mucus upward toward the pharynx in a process known as mucociliary clearance. Reflex actions such as coughing and sneezing expel trapped pathogens from the airways. In the gastrointestinal tract, peristaltic movements and defecation eliminate microorganisms. The urinary system utilizes the flushing action of urine to wash away contaminants from the urethra and bladder. Similarly, tears wash away foreign particles and microbes from the ocular surface, while saliva clears microorganisms from the oral cavity.
In the female reproductive tract, the vaginal mucosa maintains a protective ecosystem dominated by commensal organisms such as Lactobacillus species. These bacteria metabolize glycogen into lactic acid, sustaining an acidic pH that suppresses pathogenic colonization. This microbial environment acts as a biological barrier against sexually transmitted and opportunistic infections.
The gastrointestinal tract also relies on resident microbiota to maintain an anaerobic and low-pH environment that restricts pathogen survival. Competitive interactions between commensal microbes and invading organisms further reinforce colonization resistance.
Inflammation
Inflammation is a fundamental innate immune response triggered by microbial invasion, tissue injury, or cellular damage. It represents a coordinated biological reaction involving vascular changes, immune cell recruitment, and the release of soluble mediators. Both humoral components (such as complement proteins and antibodies) and cellular components (including neutrophils, macrophages, and mast cells) participate in this response.
Upon detection of infection or injury, local blood vessels undergo vasodilation, increasing blood flow to the affected site. Vascular permeability also rises, allowing plasma proteins, immune cells, and fluid to exit the circulation and accumulate in the damaged tissue. This accumulation contributes to swelling, redness, heat, and pain—classical signs of inflammation. The recruited phagocytic cells engulf and destroy invading microorganisms through phagocytosis and intracellular killing mechanisms.
Chemical mediators regulate and amplify the inflammatory response. Cytokines such as interleukins and tumor necrosis factor coordinate immune cell communication and activation. Interferons (IFNs) play a crucial role in antiviral defense by inducing an antiviral state in neighboring cells and enhancing immune surveillance. Other mediators, including prostaglandins, chemokines, and components of the complement system, contribute to pathogen recognition, leukocyte recruitment, and microbial lysis.
The inflammatory process functions to contain infections at their site of entry and prevent systemic spread. By concentrating immune effector mechanisms at the affected area, inflammation limits pathogen dissemination while promoting tissue repair and restoration of homeostasis. Although excessive inflammation may cause tissue damage, its controlled activation is essential for effective innate immune defense against bacteria, viruses, fungi, and other foreign agents.
Phagocytic Action
Phagocytic action is a fundamental component of innate immunity and is mediated by specialized immune cells collectively known as phagocytes. These cells include neutrophils, macrophages, monocytes, dendritic cells, and certain subsets of tissue-resident cells that possess the capacity to recognize, ingest, and destroy microbial pathogens and foreign particles. Upon microbial invasion or tissue injury, inflammatory mediators and chemotactic signals are released, recruiting phagocytes in large numbers to the site of infection. Through a process known as chemotaxis, these cells migrate from the bloodstream and surrounding tissues toward gradients of cytokines, complement fragments, and microbial products.
Phagocytosis refers to the active process by which phagocytes engulf extracellular particulate material such as bacteria, viruses bound to antibodies, dead cells, and debris. The process begins with pathogen recognition through pattern recognition receptors (PRRs) that detect conserved microbial structures known as pathogen-associated molecular patterns (PAMPs). Opsonization significantly enhances this recognition. Opsonins such as immunoglobulin G (IgG) and complement component C3b coat microbial surfaces and facilitate binding to specific receptors on phagocytes, thereby promoting efficient ingestion.
Following attachment, the plasma membrane of the phagocyte surrounds the target particle and internalizes it into a membrane-bound vesicle called a phagosome. The phagosome subsequently fuses with lysosomes to form a phagolysosome. Within this compartment, microorganisms are exposed to degradative enzymes, reactive oxygen species, reactive nitrogen intermediates, and acidic conditions that lead to microbial killing and digestion. This coordinated mechanism ensures rapid clearance of pathogens and contributes to containment of infection.
Cytokines
Cytokines are low–molecular weight signaling molecules that regulate immune and inflammatory responses. They are primarily produced by leukocytes, including macrophages, dendritic cells, lymphocytes, and other immune and non-immune cells in response to infection or tissue damage. Cytokines function as key communicators within the immune system, orchestrating cellular activation, differentiation, migration, and effector functions.
In innate immunity, cytokines play critical augmenting and regulatory roles during microbial invasion. Major classes of cytokines include interferons (IFNs), interleukins (ILs), tumor necrosis factor (TNF), chemokines, and colony-stimulating factors. Interferons, particularly type I interferons, are essential in antiviral defense. They induce an antiviral state in neighboring cells by upregulating genes that inhibit viral replication and enhance antigen presentation. Interleukins mediate communication between immune cells, promoting inflammation, lymphocyte activation, and differentiation. Tumor necrosis factor contributes to inflammation, fever induction, and activation of immune effector cells, while also participating in the regulation of apoptosis in infected or abnormal cells.
Cytokines also regulate the intensity and duration of inflammatory responses. Excessive cytokine production may result in systemic inflammation and pathological conditions, whereas insufficient production can impair host defense. Through tightly controlled signaling networks, cytokines ensure effective coordination between innate and adaptive immunity. In addition to antiviral activity, cytokines contribute to tumor surveillance by enhancing cytotoxic responses against transformed cells and supporting immune-mediated destruction of abnormal tissues.
Complement System
The complement system consists of a group of heat-labile serum glycoproteins that function as an essential component of innate immunity. These proteins circulate in plasma and tissue fluids in inactive forms and become sequentially activated through tightly regulated enzymatic cascades. The system is termed “complement” because it enhances or complements the activity of antibodies and other immune mechanisms during microbial invasion.
In humans, the complement system comprises nine principal components designated C1 through C9. Each component represents a specific protein that becomes activated in a sequential manner during complement cascade activation. Activation can occur through three major pathways: the classical pathway, the lectin pathway, and the alternative pathway. Although these pathways are initiated by different triggers, they converge at the activation of component C3, which serves as a central and critical molecule in complement-mediated immunity.
Complement proteins contribute to host defense through several mechanisms. Opsonization occurs when complement fragments such as C3b bind to microbial surfaces, enhancing recognition and phagocytosis by immune cells that express complement receptors. Complement activation also leads to the formation of the membrane attack complex (MAC), composed of components C5b through C9, which inserts into bacterial membranes and induces cell lysis by disrupting membrane integrity.
Furthermore, complement fragments act as potent inflammatory mediators. Molecules such as C3a and C5a function as anaphylatoxins that promote vasodilation, increase vascular permeability, and recruit leukocytes to sites of infection. C5a is particularly effective as a chemotactic factor for neutrophils and macrophages, thereby amplifying immune cell accumulation at inflammatory sites. Through these combined actions, the complement system enhances bacterial killing, promotes phagocytosis, and strengthens inflammatory responses against invading pathogens.
Complement proteins also bridge innate and adaptive immunity. Activation via the classical pathway often occurs following antigen–antibody complex formation, demonstrating how complement activity supports antibody-mediated defense. By enhancing antigen clearance and immune cell activation, the complement system reinforces the overall immune response against infection.
Mammary Glands
The mammary gland represents an anatomical structure that provides a natural protective mechanism, particularly in females during lactation. Beyond its primary role in nourishing newborns, the mammary gland contributes to immune protection through mechanical flushing and the secretion of antimicrobial components. The continuous flow of milk helps remove potential contaminants and reduces the likelihood of microbial colonization within the mammary ducts.
Breast milk contains several bioactive molecules with antimicrobial properties. Enzymes such as lysozyme degrade bacterial cell walls, thereby limiting bacterial proliferation. Other protective components include lactoferrin, immunoglobulins – especially secretory IgA – oligosaccharides, and antimicrobial peptides. These substances inhibit pathogen growth, neutralize toxins, and prevent microbial attachment to mucosal surfaces.
Colostrum, the first milk produced after childbirth, plays a particularly important immunological role. It is enriched with high concentrations of antibodies, immune cells, cytokines, and growth factors that provide passive immunity to the newborn. Since the neonatal immune system is immature at birth, colostrum supplies immediate protection against environmental pathogens and supports the development of the infant’s immune defenses.
Through its mechanical flushing action and secretion of immune-active molecules, the mammary gland contributes to early-life immune protection and reduces the risk of infections in both mother and child.
REFERENCES
Abbas A.K, Lichtman A.H and Pillai S (2010). Cellular and Molecular Immunology. Sixth edition. Saunders Elsevier Inc, USA.
Actor J (2014). Introductory Immunology. First edition. Academic Press, USA.
Alberts B, Bray D, Johnson A, Lewis J, Raff M, Roberts K and Walter P (1998). Essential Cell Biology: An Introduction to the Molecular Biology of the Cell. Third edition. Garland Publishing Inc., New York.
Bach F and Sachs D (1987). Transplantation immunology. N. Engl. J. Med. 317(8):402-409.
Barrett J.T (1998). Microbiology and Immunology Concepts. Philadelphia, PA: Lippincott-Raven Publishers. USA.
Jaypal V (2007). Fundamentals of Medical Immunology. First edition. Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India.
John T.J and Samuel R (2000). Herd Immunity and Herd Effect: New Insights and Definitions. European Journal of Epidemiology, 16:601-606.
Levinson W (2010). Review of Medical Microbiology and Immunology. Twelfth edition. The McGraw-Hill Companies, USA.
Roitt I, Brostoff J and Male D (2001). Immunology. Sixth edition. Harcourt Publishers Limited, Spain.
Zon LI (1995). Developmental biology of hematopoiesis. Blood, 86(8): 2876–91.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.