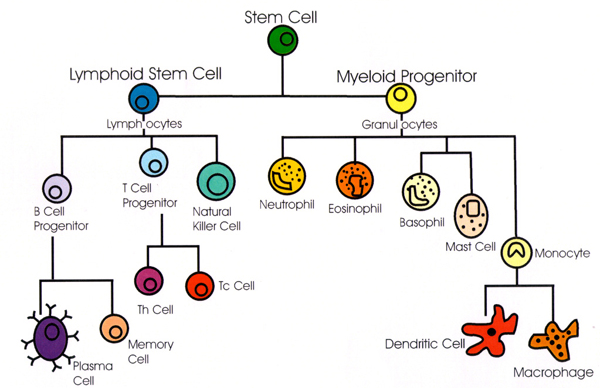
All blood and immune cells originate from hematopoietic stem cells within the bone marrow through the tightly regulated process of hematopoiesis. Hematopoiesis is defined as the biological mechanism responsible for the continuous formation, differentiation, and maturation of blood cellular components, including erythrocytes, leukocytes, and thrombocytes, as well as specialized immune cells such as B and T lymphocytes. This process ensures the maintenance of blood homeostasis and sustained immune competence throughout the lifespan of an organism.
Hematopoietic stem cells (HSCs) reside primarily in the medullary cavity of bones, particularly within specialized microenvironments known as stem cell niches. These niches provide structural support and molecular signals that regulate self-renewal and lineage commitment. HSCs possess the dual capacity for self-renewal and multipotent differentiation, enabling them to generate all major blood cell lineages. Under physiological conditions, hematopoiesis is a continuous process that compensates for the short lifespan of circulating blood cells and responds dynamically to infection, inflammation, hypoxia, or blood loss.
During hematopoiesis, hematopoietic stem cells first differentiate into multipotent progenitor cells, which subsequently give rise to distinct lineage-committed progenitors. These progenitors include erythroid progenitor cells that develop into red blood cells responsible for oxygen transport; myeloid progenitor cells that produce granulocytes, monocytes, macrophages, and platelets; and lymphoid progenitor cells that generate adaptive immune cells such as B lymphocytes, T lymphocytes, and natural killer cells. The differentiation process is tightly controlled by transcription factors, cytokines, growth factors, and intracellular signaling pathways that guide lineage specification and maturation.
Hematopoiesis occurs predominantly in the bone marrow in adults, whereas in fetal development it initially takes place in the yolk sac, followed by the liver and spleen before transitioning to the bone marrow. Disruptions in this process can lead to hematological disorders such as anemia, immunodeficiency, leukemia, and other bone marrow failure syndromes. Therefore, proper regulation of hematopoiesis is essential for maintaining immune function and overall physiological stability.
Hematopoietic stem and progenitor cells differentiate into distinct lineages that give rise to the major cellular components of blood. The erythroid lineage undergoes proliferation and maturation to produce erythrocytes, commonly known as red blood cells (RBCs), which are specialized for oxygen transport, and reticulocytes, which represent immature RBCs that still retain residual ribosomal RNA and continue to mature after release into the circulation. These cells are essential for maintaining tissue oxygenation and metabolic homeostasis. In contrast, the myeloid lineage differentiates into myeloid precursor cells that further develop into diverse cell types, including macrophages, granulocytes (such as neutrophils, eosinophils, and basophils), and megakaryocytes. Macrophages and granulocytes serve key functions in innate immune defense through phagocytosis, cytokine production, and rapid responses to invading pathogens. Megakaryocytes are responsible for the production of platelets, which play a critical role in hemostasis and also contribute to immune modulation and inflammatory responses. The lymphoid lineage primarily gives rise to lymphocytes, which include T lymphocytes and B lymphocytes, constituting major components of the adaptive immune system and circulating as white blood cells (WBCs). T cells mediate cell-mediated immunity and regulate immune responses, whereas B cells are responsible for antibody production and humoral immunity.
The immune system is an integrated and dynamic network composed of specialized cells, tissues, and soluble mediators that function collaboratively to defend the host against infectious agents and pathogenic microorganisms. It includes primary and secondary lymphoid organs, immune effector cells, and immunological molecules such as antibodies, cytokines, and complement proteins. These components interact through tightly regulated signaling pathways to detect, neutralize, and eliminate pathogens while maintaining tolerance to self-antigens. In all living animals, including humans, this complex defense architecture is fundamental for survival, as it provides protection against microbial invasion, tissue damage, and disease progression. Through coordinated innate and adaptive mechanisms, the immune system prevents colonization by pathogens and limits the spread of infection, thereby preserving physiological integrity and overall health.
Immune System Cells and Host Defense Mechanisms
Immune system cells are distributed throughout the body, residing in tissues, circulating in blood and lymph, and positioned at barrier surfaces such as the skin, respiratory tract, and gastrointestinal lining. Their strategic localization enables rapid detection and coordinated responses to invading antigens and pathogenic microorganisms. Upon recognition of foreign molecules, these cells initiate a cascade of innate and adaptive immune reactions aimed at neutralizing, eliminating, or containing the threat. Through tightly regulated signaling networks and cellular interactions, they preserve physiological balance and protect host integrity.
The immune system represents the primary biological defense mechanism against infectious diseases. It functions through an integrated network of cellular and molecular components that identify non-self structures and mount targeted responses. When immune competence is intact, pathogens that breach physical barriers are rapidly detected and cleared before they establish infection. However, immune function can be impaired by factors such as malnutrition, chronic psychological or physical stress, genetic disorders, immunosuppressive therapies, and debilitating infections including human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS). In such conditions, immune surveillance is weakened, allowing opportunistic and pathogenic organisms to invade, replicate, and cause disease.
The effectiveness of host immunity is therefore critical in preventing microbial invasion and subsequent colonization of tissues. A functional immune system limits pathogen dissemination through coordinated actions involving antigen recognition, phagocytosis, cytotoxic responses, antibody production, and inflammatory signaling. Failure in any of these mechanisms increases susceptibility to recurrent or severe infections. Immunological competence also contributes to immune memory, ensuring faster and more efficient responses upon re-exposure to previously encountered pathogens.
Multiple specialized cell populations contribute to immune defense. Lymphoid cells, including lymphocytes, play central roles in adaptive immunity. B lymphocytes differentiate into plasma cells that produce antibodies and memory B cells that provide long-term protection. T lymphocytes mediate cellular immune responses, assisting other immune cells or directly destroying infected cells. Phagocytic cells such as macrophages and neutrophils engulf and degrade invading microorganisms while simultaneously presenting antigens to activate adaptive responses. Dendritic cells function as professional antigen-presenting cells that bridge innate and adaptive immunity by capturing antigens and initiating T-cell activation.
Additional immune cell types include monocytes, which circulate in blood and differentiate into macrophages or dendritic cells in tissues; granulocytes such as basophils and eosinophils, which participate in inflammatory and allergic responses; and megakaryocytes, which give rise to platelets involved in hemostasis and inflammatory signaling. Null cells and natural killer (NK)–like populations contribute to early defense by targeting infected or transformed cells without prior sensitization. Collectively, these cells operate through dynamic communication mediated by cytokines, chemokines, and surface receptor interactions. Together, the coordinated activity of these immune components ensures continuous surveillance and rapid mobilization against pathogens, maintaining host homeostasis and reducing disease burden.
Types of Immune System Cells
- Lymphocytes (B cells and T cells)
- Memory B cells
- Plasma cells
- Phagocytes
- Macrophages
- Monocytes
- Dendritic cells
- Granulocytes
- Neutrophils
- Eosinophils
- Basophils
- Megakaryocytes
- Null cells
LYMPHOCYTES
Lymphocytes are mononuclear leukocytes that play a central role in coordinating both humoral (antibody-mediated) and cell-mediated immune responses. They constitute a major cellular component of the adaptive immune system and are essential for targeted and regulated host defense. The principal lymphocyte subsets include B lymphocytes (B cells), T lymphocytes (T cells), and natural killer (NK) cells. Among these, B and T cells are the hallmark cells of adaptive immunity, whereas NK cells are generally classified as innate lymphoid cells with functions that bridge innate and adaptive immune mechanisms.
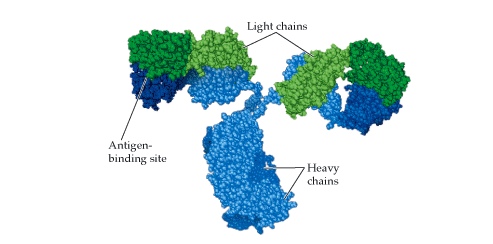
B and T lymphocytes often collectively referred to as lymphoid cells are critical mediators of immune specificity and memory. These cells underpin the defining characteristics of adaptive immunity, including the ability to distinguish self from non-self antigens, antigen specificity, clonal expansion, receptor diversity, and the generation of long-lived immunological memory. Through highly specific antigen receptor rearrangement, B cells express membrane-bound immunoglobulins that function as antigen receptors and, upon activation, differentiate into plasma cells that secrete antibodies. These antibodies circulate systemically and neutralize pathogens, facilitate opsonization, and activate complement pathways, thereby mediating humoral immunity.
T lymphocytes are primarily responsible for cell-mediated immune responses. They differentiate into distinct functional subsets, such as helper T cells (CD4⁺), which coordinate immune activation through cytokine secretion, and cytotoxic T cells (CD8⁺), which directly eliminate infected or aberrant cells. Regulatory T cells further contribute to immune homeostasis by suppressing excessive or autoreactive responses. Collectively, these subsets ensure precise regulation and effective immune surveillance.
The nomenclature of B and T lymphocytes reflects their anatomical sites of maturation. B cells derive their name from the Bursa of Fabricius in birds, the primary lymphoid organ responsible for B-cell development in avian species, and from the bone marrow in mammals and humans, where B-cell maturation occurs. T cells are so designated because they mature in the thymus, a specialized primary lymphoid organ that supports T-cell development and selection. These developmental origins underscore the functional specialization and evolutionary conservation of adaptive immune architecture.
B Cells
B lymphocytes are central mediators of humoral immunity and are uniquely equipped to recognize native antigens in their soluble or particulate forms through membrane-bound immunoglobulins known as B cell receptors (BCRs). Unlike T lymphocytes, B cells do not require antigen processing and presentation by major histocompatibility complex (MHC) molecules for initial antigen recognition. Upon direct binding of antigen to the BCR, and with appropriate costimulatory signals often provided by helper T cells – B cells become activated and undergo clonal expansion and differentiation. The primary effector outcome of this activation is differentiation into plasma cells that secrete large quantities of antigen-specific antibodies. These antibodies neutralize pathogens, opsonize microbes to enhance phagocytosis, activate the complement cascade, and mediate antibody-dependent cellular cytotoxicity.
B cells originate and undergo early maturation in the bone marrow, where processes such as V(D)J recombination generate receptor diversity and central tolerance mechanisms eliminate strongly self-reactive clones. Following maturation, naïve B cells circulate through secondary lymphoid organs where antigen encounter occurs. After activation, some B cells differentiate into short-lived plasma cells that provide immediate antibody production, whereas others participate in germinal center reactions to undergo affinity maturation and class-switch recombination, enhancing antibody specificity and functional versatility. A critical subset also differentiates into memory B cells, which persist in a quiescent state and enable rapid, robust secondary responses upon re-exposure to the same antigen. B cells constitute the primary cellular basis of antibody-mediated immunity and contribute to long-term immunological memory and rapid protective responses against previously encountered pathogens.
T Cells
T lymphocytes are key effectors of cell-mediated immunity and require antigen recognition in the context of major histocompatibility complex (MHC) molecules. Unlike B cells, T cells cannot recognize free antigens; instead, their T cell receptors (TCRs) detect processed peptide fragments presented on the surface of antigen-presenting cells. Antigen presentation through MHC class I molecules activates cytotoxic T cells (CD8⁺), whereas peptide presentation via MHC class II molecules activates helper T cells (CD4⁺). T cells originate from hematopoietic stem cells in the bone marrow but undergo maturation and selection within the thymus. During thymic development, T cells express diverse TCRs generated through somatic recombination, followed by positive and negative selection to ensure self-MHC recognition while eliminating strongly self-reactive clones. Mature T cells then migrate to peripheral lymphoid tissues as naïve cells ready for antigen encounter.
Upon activation, T cells proliferate and differentiate into specialized effector subsets. CD8⁺ cytotoxic T lymphocytes directly eliminate infected, malignant, or allogeneic target cells through mechanisms such as perforin-mediated lysis and induction of apoptosis via granzymes and death receptor pathways. They also contribute to immune regulation by modulating immune responses. CD4⁺ helper T cells orchestrate adaptive immunity by secreting cytokines that regulate and enhance the functions of B cells, macrophages, dendritic cells, and other lymphocytes. Distinct helper subsets, including Th1, Th2, Th17, and regulatory T cells, further specialize in coordinating inflammatory responses, antibody production, and immune tolerance. T cells are indispensable for targeted immune defense, immune regulation, and coordination of adaptive immune responses.
As aforesaid, immune system cells develop through a process known as hematopoiesis in the bone marrow (Figure 1).
Phagocytes
Phagocytes are immune cells primarily responsible for recognizing, engulfing, and eliminating microbial pathogens, particularly bacteria, through a process known as phagocytosis. During phagocytosis, these cells internalize foreign microorganisms into intracellular vesicles called phagosomes, which subsequently fuse with lysosomes where enzymatic degradation and oxidative mechanisms destroy the engulfed material. This function is essential for limiting infection and maintaining tissue homeostasis. Key examples of phagocytic cells include macrophages, neutrophils, and eosinophils, each playing distinct but complementary roles in innate immune defense.
Macrophages reside in tissues and act as sentinel cells that detect pathogens, remove cellular debris, and present antigens to adaptive immune cells, thereby linking innate and adaptive immunity. Neutrophils are rapidly recruited to sites of infection and serve as the first line of cellular defense, exhibiting strong microbicidal activity. Eosinophils contribute to defense against parasitic infections and modulate inflammatory responses.
The overall effectiveness of host immunity depends on the coordinated activity of diverse immune cell populations. These include lymphocytes, dendritic cells, monocytes, plasma cells, memory B cells, granulocytes, megakaryocytes, basophils, eosinophils, neutrophils, and natural killer cells. Upon recognition of invading antigens, these cells are mobilized to restore physiological balance and prevent microbial colonization and disease progression.
Macrophages
Macrophages are highly versatile immune cells that perform essential roles in both innate and adaptive immunity. They function as professional antigen-presenting cells by processing and presenting antigens to T lymphocytes, thereby linking innate immune detection to adaptive immune activation. In addition to antigen presentation, macrophages exhibit potent phagocytic activity, enabling them to engulf and degrade pathogens, cellular debris, and apoptotic cells. They also secrete a broad spectrum of cytokines and chemokines that regulate inflammation, coordinate immune cell recruitment, and modulate tissue repair. Furthermore, macrophages contribute to antibody-dependent cell-mediated cytotoxicity (ADCC), enhancing the elimination of opsonized targets.
Macrophages are distributed throughout virtually all tissues and adapt their phenotype according to the local microenvironment. Tissue-resident macrophage populations include Kupffer cells in the liver and histiocytes in connective tissues, both of which contribute to immune surveillance and homeostasis. In the lungs, alveolar macrophages defend against inhaled pathogens, while mesangial cells in the kidneys perform macrophage-like immune and structural functions. In the central nervous system, microglial cells serve as resident immune regulators, and osteoclasts in bone participate in tissue remodeling while retaining macrophage lineage characteristics. Collectively, these specialized populations underscore the functional diversity and systemic importance of macrophages.
Dendritic Cells
Dendritic cells (DCs) are specialized immune cells that function primarily as professional antigen-presenting cells (APCs), playing a central role in initiating and regulating immune responses. A major subset includes Langerhans cells, which are located in the epidermis beneath the skin and act as sentinels against invading pathogens. DCs bridge innate and adaptive immunity by capturing, processing, and presenting antigens to lymphocytes, thereby triggering antigen-specific immune activation.
These cells are widely distributed throughout the body and are found in peripheral blood, spleen, lymph nodes, thymus, and various tissues where immune surveillance occurs. Like macrophages, dendritic cells are considered professional APCs due to their high efficiency in antigen uptake and presentation. However, they are uniquely specialized for T-cell activation.
Dendritic cells process internalized antigens into peptide fragments and present them on major histocompatibility complex (MHC) class II molecules. This enables recognition by CD4+ T helper cells through T-cell receptors, leading to T-cell activation, differentiation, and subsequent immune responses. Through costimulatory molecule expression and cytokine secretion, dendritic cells regulate immune tolerance and immunity, making them essential components of host defense and immunological homeostasis.
Memory B Cells
Memory B cells are a specialized subset of B lymphocytes that persist in the circulation and secondary lymphoid tissues after an initial immune response. They remain functionally quiescent yet viable for extended periods, providing long-term immunological surveillance. Upon re-exposure to a previously encountered pathogen or antigen, memory B cells are rapidly reactivated, enabling a swift and robust secondary immune response.
During primary infection, antigen stimulation triggers the differentiation of naïve B cells into effector plasma cells, which produce antibodies, and into memory B cells that are retained for future protection. The generation of memory cells represents an essential outcome of adaptive immunity, ensuring enhanced speed and magnitude of response upon subsequent antigen encounters. These cells circulate through the bloodstream and lymphatic system, continuously monitoring for cognate antigens.
When reactivated, memory B cells proliferate rapidly and differentiate into antibody-secreting plasma cells with higher affinity for the antigen, owing to prior affinity maturation and class switching. This accelerated response limits pathogen replication and contributes to immunological memory. Together with memory T cells, memory B cells form the cellular basis for long-term protective immunity and vaccine-induced defense.
Plasma Cells
Plasma cells are differentiated antibody-secreting effector cells derived from activated immunocompetent B lymphocytes following antigen stimulation. Upon recognition of a specific antigen and subsequent activation, B cells proliferate and undergo differentiation into plasma cells, which function as the principal producers of immunoglobulins. Although plasma cells are generally short-lived in peripheral circulation and tissues, they exhibit a highly specialized capacity to synthesize and secrete large quantities of antibodies within their limited lifespan.
These cells generate immunoglobulins that are highly specific for antigenic determinants, also known as epitopes, present on invading pathogens or foreign substances. The secreted antibodies bind selectively to their corresponding antigens, thereby neutralizing toxins, opsonizing pathogens for phagocytosis, and activating complement-mediated immune responses. This targeted antibody production is essential for adaptive humoral immunity and contributes to immune memory and long-term protection.
Some plasma cells migrate to survival niches in the bone marrow or secondary lymphoid organs, where they may persist for extended periods and continue antibody secretion. Through rapid and sustained immunoglobulin production, plasma cells play a critical role in controlling infections and maintaining immune defense mechanisms.
Granulocytes
Granulocytes are a major class of white blood cells (leukocytes) characterized by the presence of cytoplasmic granules that contain enzymes and antimicrobial substances. These granules play a critical role in immune defense by facilitating pathogen recognition, destruction, and regulation of inflammatory responses. The principal types of granulocytes include neutrophils, eosinophils, and basophils, each with distinct functional roles in host protection. Neutrophils are the most abundant and act as first responders to bacterial and fungal infections through phagocytosis and release of reactive oxygen species. Eosinophils are primarily involved in defense against parasitic infections and in modulating allergic inflammation. Basophils contribute to inflammatory and hypersensitivity reactions by releasing histamine and other mediators.
Granulocytes differ from agranulocytes, such as monocytes and lymphocytes, which lack prominent cytoplasmic granules and perform different immune functions. These cells are produced in the bone marrow through a process called granulopoiesis and are released into the bloodstream to circulate and migrate to sites of infection or tissue injury. Upon activation, granulocytes rapidly respond to invading pathogens or antigens by engaging in phagocytosis, degranulation, and cytokine secretion, thereby contributing to innate immunity and inflammatory regulation.
Megakaryocytes
Megakaryocytes are large, polyploid cells derived from hematopoietic stem and progenitor cells and primarily located in the bone marrow. They undergo a unique process of endomitosis, resulting in multiple nuclei and extensive cytoplasmic expansion. Their distinctive morphology and genomic amplification enable them to generate and release large numbers of platelets into the circulation. Platelets, also known as thrombocytes, play a central role in hemostasis by mediating blood clot formation and contributing to vascular repair following injury.
The differentiation and maturation of megakaryocytes are tightly regulated by growth factors and cytokines, particularly thrombopoietin, which is the principal regulator of megakaryopoiesis. During maturation, megakaryocytes develop an elaborate internal membrane system and extend long cytoplasmic projections called proplatelets into the bone marrow sinusoids. Fragmentation of these projections leads to the release of functional platelets into the bloodstream.
Beyond platelet production, emerging evidence suggests that megakaryocytes may contribute to the regulation of the bone marrow microenvironment and participate in immune modulation. Dysregulation of megakaryocyte development or function can result in abnormal platelet counts and is associated with hematological disorders such as thrombocytopenia and certain myeloproliferative diseases.
Basophils
Basophils are rare, granulated white blood cells (leukocytes) that play a key role in immune regulation and hypersensitivity responses. Their cytoplasm contains bioactive mediators such as histamine, a vasoactive amine released during allergic and inflammatory reactions, and heparin, an anticoagulant that modulates blood clotting and vascular permeability. Basophils are primarily located in the peripheral blood circulation and are rapidly recruited to sites of tissue injury or antigen exposure to participate in early inflammatory responses.
In allergic reactions, basophils are activated when antigens bind to immunoglobulin E (IgE) antibodies that are attached to high-affinity Fc receptors on their surface. Cross-linking of IgE–antigen complexes triggers cellular degranulation, resulting in the release of histamine and other pro-inflammatory mediators such as leukotrienes and cytokines. This process contributes to vasodilation, increased vascular permeability, smooth muscle contraction, and the clinical manifestations of hypersensitivity reactions. Laboratory identification of basophils is based on their morphological features and staining characteristics. They stain dark blue or purple with basic dyes due to their granule content, which aids in differentiation from other granulocytes during microscopic examination of blood smears.
Eosinophils
Eosinophils are a specialized subset of granulocytes characterized by the presence of cytoplasmic granules that readily absorb eosin, a red acidic dye used in histological staining. Under microscopic examination, these cells appear distinctly red or reddish-orange due to their affinity for eosin. They originate from hematopoietic stem cells in the bone marrow and circulate in peripheral blood before migrating into tissues where they exert effector functions.
Functionally, eosinophils contribute to innate and adaptive immune responses. Although traditionally associated with host defense against parasitic infections, particularly helminths and nematodes, they also participate in modulating inflammatory reactions and immune regulation. Eosinophils possess phagocytic capabilities and can engulf pathogens, immune complexes, and cellular debris through phagocytosis. In addition, they release cytotoxic granule proteins, reactive oxygen species, and cytokines that enhance antimicrobial activity and influence other immune cells.
Beyond infection, eosinophils are implicated in allergic conditions and chronic inflammatory diseases, where their excessive activation may contribute to tissue damage. Their functional versatility highlights their importance in immune surveillance, pathogen clearance, and regulation of inflammatory processes within tissues.
Neutrophils
Neutrophils are a class of granulocytic leukocytes that play a central role in innate immunity through rapid phagocytic and inflammatory responses. They constitute the most abundant circulating white blood cell population and act as first responders to sites of infection or tissue injury. Morphologically, neutrophils are characterized by multilobed nuclei and cytoplasmic granules. When stained with Wright’s stain, they appear neutral to pale pink due to the staining properties of their granules.
Neutrophils are also referred to as polymorphonuclear leukocytes (PMNs) because of their segmented nuclei. Functionally, they are widely recognized as bacteria-eating granulocytes owing to their strong chemotactic migration toward microbial signals and their capacity for efficient phagocytosis. Upon encountering pathogens, neutrophils engulf and destroy them using antimicrobial mechanisms that include the production of reactive oxygen species, proteolytic enzymes, and antimicrobial peptides. They can also release neutrophil extracellular traps (NETs), which immobilize and neutralize invading microorganisms. Neutrophils are bactericidal effector cells that provide rapid, non-specific defense and contribute significantly to early host protection against infection.
Null Cells
Null cells refer to immature B lymphocytes that have not yet encountered or been activated by a specific antigen. These cells represent an early stage in B-cell development and circulate in the peripheral blood and lymphoid tissues in a resting state. They are often described as naïve B cells because they have not undergone antigen-driven differentiation, clonal expansion, or functional specialization. In immunological classification, the term “null” historically described lymphocytes that lacked definitive surface markers distinguishing them as either T or B cells; however, it is now commonly associated with immature or undifferentiated B-cell populations.
Null or naïve B cells express surface immunoglobulin receptors that enable antigen recognition but have not yet been exposed to their cognate antigen. Upon antigen binding and appropriate co-stimulatory signals, these cells become activated, proliferate, and differentiate into plasma cells or memory B cells. Plasma cells produce antigen-specific antibodies, while memory B cells provide long-term immune protection. Functionally, null cells are essential for maintaining immune readiness, ensuring that the adaptive immune system retains a diverse repertoire capable of responding to novel pathogens. Their proper development and regulation are critical for effective immune surveillance and immune homeostasis.
Monocytes
Monocytes are large, nucleated leukocytes that play a central role in the innate immune response. They originate from hematopoietic stem cells in the bone marrow and circulate in the bloodstream before migrating into tissues where they differentiate into macrophages and dendritic cells. A key function of monocytes is phagocytosis, whereby they recognize, engulf, and degrade invading pathogens, including bacteria, viruses, and other foreign particles. This activity is initiated upon detection of antigens or pathogen-associated molecular patterns through pattern recognition receptors on their surface.
Beyond direct pathogen clearance, monocytes contribute to immune regulation by producing cytokines and chemokines that modulate inflammatory responses and recruit additional immune cells to sites of infection or tissue injury. They also participate in antigen presentation, linking innate and adaptive immunity by processing and displaying antigens to T lymphocytes. Monocytes are heterogeneous, comprising subsets with distinct functional properties that influence inflammation, tissue repair, and immune surveillance. Through these coordinated activities, monocytes serve as essential components of host defense, maintaining immune homeostasis and responding rapidly to infectious and inflammatory stimuli.
REFERENCES
Abbas A.K, Lichtman A.H and Pillai S (2010). Cellular and Molecular Immunology. Sixth edition. Saunders Elsevier Inc, USA.
Actor J (2014). Introductory Immunology. First edition. Academic Press, USA.
Alberts B, Bray D, Johnson A, Lewis J, Raff M, Roberts K and Walter P (1998). Essential Cell Biology: An Introduction to the Molecular Biology of the Cell. Third edition. Garland Publishing Inc., New York.
Bach F and Sachs D (1987). Transplantation immunology. N. Engl. J. Med. 317(8):402-409.
Barrett J.T (1998). Microbiology and Immunology Concepts. Philadelphia, PA: Lippincott-Raven Publishers. USA.
Jaypal V (2007). Fundamentals of Medical Immunology. First edition. Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India.
John T.J and Samuel R (2000). Herd Immunity and Herd Effect: New Insights and Definitions. European Journal of Epidemiology, 16:601-606.
Levinson W (2010). Review of Medical Microbiology and Immunology. Twelfth edition. The McGraw-Hill Companies, USA.
Roitt I, Brostoff J and Male D (2001). Immunology. Sixth edition. Harcourt Publishers Limited, Spain.
Zon LI (1995). Developmental biology of hematopoiesis. Blood, 86(8): 2876–91.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.