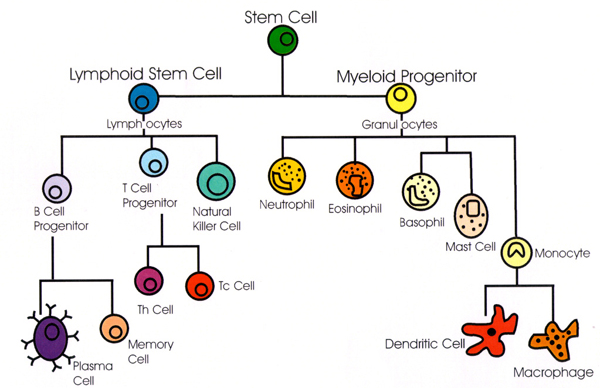
Innate immunity constitutes the evolutionarily conserved first line of host defense and is present across virtually all multicellular organisms. Unlike adaptive immunity, which requires clonal expansion and antigen-specific priming, innate immune responses are immediate and operate at near-maximal capacity upon exposure to foreign material. This rapid reactivity is mediated by germline-encoded pattern recognition receptors (PRRs) that detect conserved pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs), triggering inflammation, cytotoxicity, and phagocytosis. The principal cellular effectors of innate immunity include natural killer (NK) cells, professional phagocytes, and cells of the reticuloendothelial (mononuclear phagocyte) system. NK cells are cytotoxic lymphocytes that eliminate virus-infected and transformed cells through perforin- and granzyme-dependent mechanisms, while also producing immunomodulatory cytokines such as interferon-γ (IFN-γ).
Their activity is regulated by a balance of activating and inhibitory receptors that assess target cell expression of major histocompatibility complex (MHC) class I molecules. Phagocytic cells – primarily neutrophils, monocytes, macrophages, and dendritic cells ingest and destroy invading microorganisms through oxidative burst, lysosomal degradation, and antimicrobial peptide release. These cells also orchestrate inflammation via cytokine and chemokine secretion and serve as a bridge to adaptive immunity through antigen presentation. The reticuloendothelial system comprises tissue-resident macrophages and mononuclear phagocytes distributed in organs such as the liver (Kupffer cells), spleen, lymph nodes, and bone marrow. These cells filter blood and lymph, clear immune complexes and apoptotic debris, and maintain tissue homeostasis. Innate immune cells provide rapid pathogen containment, initiate inflammatory signaling cascades, and shape downstream adaptive immune responses, thereby establishing a critical foundation for effective host defense.
NATURAL KILLER (NK) CELLS
Natural killer (NK) cells are large granular lymphocytes that constitute a critical component of the innate immune system. They circulate in peripheral blood and are also found in secondary lymphoid organs and peripheral tissues. NK cells are primarily responsible for the rapid identification and elimination of virally infected cells, transformed (tumor) cells, and certain intracellular pathogens. Unlike B and T lymphocytes, NK cells do not require prior antigen sensitization to exert cytotoxic activity, enabling them to respond early during infection or malignant transformation. Morphologically, NK cells are characterized by prominent cytoplasmic granules containing cytotoxic effector molecules, notably perforin and granzymes. Upon recognition of a susceptible target cell, NK cells form an immunological synapse and release these granule contents in a directed manner. Perforin polymerizes within the target cell membrane to form pores, facilitating the entry of granzymes—serine proteases that activate intracellular caspase cascades. This process induces apoptosis (programmed cell death) in the target cell, thereby limiting viral replication and preventing the proliferation of malignant cells. In addition to granule-mediated cytotoxicity, NK cells can induce apoptosis through death receptor pathways, such as Fas ligand (FasL) and TRAIL interactions.
Functionally, NK cells are defined as non-B, non-T lymphocytes because they do not express rearranged antigen-specific receptors such as the B cell receptor (BCR) or T cell receptor (TCR). In non-immunized individuals, NK cells provide major histocompatibility complex (MHC)-dependent cytolytic activity, particularly against cells with altered or diminished expression of MHC class I molecules. This feature underpins the “missing-self” recognition model. Many viruses and tumor cells downregulate MHC class I expression to evade cytotoxic T lymphocyte detection; however, this renders them more susceptible to NK cell-mediated killing. NK cell activity is tightly regulated by a balance between activating and inhibitory surface receptors. These include killer activation receptors (e.g., NKG2D and certain natural cytotoxicity receptors) and killer inhibitory receptors (e.g., killer-cell immunoglobulin-like receptors, KIRs, and CD94/NKG2A). Inhibitory receptors recognize self MHC class I molecules expressed on healthy cells. Engagement of these receptors transmits dominant negative signals that suppress NK cell activation, thereby preventing damage to normal tissues. Conversely, when MHC class I expression is reduced or absent, inhibitory signaling is diminished, and activating receptor engagement by stress-induced ligands or viral glycoproteins triggers cytotoxic degranulation and cytokine production.
Beyond direct cytotoxicity, NK cells are potent immunoregulatory cells. Upon activation, they secrete a range of cytokines and chemokines, including interferon-gamma (IFN-γ), tumor necrosis factor (TNF), and various interleukins. These mediators enhance macrophage activation, promote Th1-type adaptive immune responses, and contribute to the orchestration of inflammation. Although traditionally categorized as innate immune cells lacking immunological memory, emerging evidence suggests that certain NK cell subsets can exhibit memory-like responses under specific conditions. NK cells are circulating effector lymphocytes that provide rapid, antigen-independent defense against viral infections and malignancies through granule-mediated cytotoxicity, death receptor signaling, and cytokine production, all regulated by a finely tuned balance of activating and inhibitory receptor interactions with MHC class I molecules.
PHAGOCYTES
Phagocytes are specialized effector cells of the innate immune system that detect, internalize, and eliminate invading microorganisms, apoptotic cells, and particulate debris. They constitute a central arm of host defense and tissue homeostasis by executing phagocytosis – a tightly regulated, multistep process involving recognition, engulfment, intracellular killing, and degradation of targets. Beyond simple microbial clearance, phagocytes orchestrate inflammatory responses, shape adaptive immunity, and contribute to tissue repair. Phagocytosis begins with target recognition. Phagocytes express an array of pattern recognition receptors (PRRs), including Toll-like receptors (TLRs), C-type lectin receptors, scavenger receptors, and complement receptors. These receptors bind conserved microbial motifs known as pathogen-associated molecular patterns (PAMPs), such as lipopolysaccharide (LPS), peptidoglycan, or β-glucans. Recognition is often enhanced through opsonization, in which pathogens are coated with antibodies (IgG) or complement components (e.g., C3b), facilitating engagement via Fc receptors or complement receptors and improving phagocytic efficiency.
Following receptor engagement, the phagocyte membrane extends pseudopodia around the target, forming an intracellular vesicle termed a phagosome. The phagosome subsequently fuses with lysosomes to generate a phagolysosome, where the ingested material is subjected to a hostile microenvironment characterized by acidification, hydrolytic enzymes, antimicrobial peptides, and reactive intermediates. Neutrophils and macrophages generate reactive oxygen species (ROS) through the NADPH oxidase complex and reactive nitrogen species (RNS) via inducible nitric oxide synthase (iNOS), creating oxidative stress that is lethal to many pathogens. Proteases, defensins, lysozyme, and other lysosomal enzymes further degrade microbial structures. Phagocytes also function as immunomodulatory cells. Macrophages and dendritic cells process internalized antigens and present peptide fragments via major histocompatibility complex (MHC) molecules to T lymphocytes, linking innate and adaptive immunity. In addition, activated phagocytes secrete cytokines and chemokines (e.g., TNF-α, IL-1β, IL-6, CXCL8) that recruit additional immune cells, amplify inflammation, and coordinate systemic responses such as fever and acute-phase protein production.
Several leukocyte subsets possess phagocytic capacity. Neutrophils are short-lived, highly abundant granulocytes that serve as rapid first responders to infection. They exhibit potent microbicidal activity, including degranulation and the formation of neutrophil extracellular traps (NETs). Macrophages, derived from circulating monocytes or resident in tissues (e.g., Kupffer cells in the liver, alveolar macrophages in the lung), provide sustained phagocytic activity, immune surveillance, and regulation of inflammation. They display functional plasticity, broadly categorized into pro-inflammatory (M1-like) and anti-inflammatory or tissue-repair (M2-like) activation states. Eosinophils, although primarily associated with helminth defense and allergic inflammation, also possess limited phagocytic activity and release cytotoxic granule proteins. Dendritic cells share phagocytic properties but are particularly specialized for antigen presentation. It is important to distinguish phagocytes from other innate lymphoid cells such as natural killer (NK) cells. While NK cells contribute to early antimicrobial defense through cytotoxicity and cytokine production, they eliminate infected host cells rather than engulfing pathogens and therefore do not perform classical phagocytosis. Phagocytes are indispensable components of innate immunity. Through coordinated recognition systems, intracellular killing mechanisms, and immunoregulatory signaling, they provide immediate protection against infection while simultaneously directing the development of adaptive immune responses and maintaining tissue integrity.
NEUTROPHILS, EOSINOPHILS and BASOPHILS
Neutrophils
Neutrophil are the most abundant circulating leukocytes in human peripheral blood, typically comprising 50-70% of total white blood cells. They are terminally differentiated, short-lived granulocytes characterized by a multilobed (polymorphonuclear) nucleus and cytoplasm packed with azurophilic (primary) and specific (secondary) granules. These granules contain a broad repertoire of antimicrobial effectors, including myeloperoxidase, defensins, cathepsins, lysozyme, lactoferrin, and proteases such as elastase. Functionally, neutrophils are central to innate immunity and represent the first line of cellular defense against invading microorganisms, particularly extracellular bacteria and fungi. They are rapidly recruited to sites of infection or tissue injury in response to chemotactic signals such as IL-8 (CXCL8), complement fragments (C5a), leukotriene B4, and bacterial formyl peptides. Following adhesion to activated endothelium and extravasation into tissues, neutrophils eliminate pathogens primarily through phagocytosis. Engulfed microbes are sequestered within phagosomes, which fuse with granules to form phagolysosomes where oxidative and non-oxidative killing mechanisms operate.
The oxidative burst, mediated by the NADPH oxidase complex, generates reactive oxygen species (ROS) including superoxide anion and hydrogen peroxide; myeloperoxidase subsequently catalyzes the formation of hypochlorous acid, a potent microbicidal agent. In addition to intracellular killing, neutrophils can release neutrophil extracellular traps (NETs) -web-like chromatin structures coated with antimicrobial proteins that immobilize and neutralize pathogens extracellularly. Beyond direct antimicrobial action, neutrophils modulate inflammation by secreting cytokines, chemokines, and lipid mediators that amplify or resolve immune responses. Dysregulation of neutrophil function contributes to pathology, including chronic inflammatory diseases, acute respiratory distress syndrome, and tissue damage associated with excessive ROS and protease release. Conversely, quantitative or functional neutrophil deficiencies (e.g., neutropenia or chronic granulomatous disease) markedly increase susceptibility to infection. Thus, neutrophils are indispensable effectors of innate host defense and key orchestrators of early inflammatory responses.
Eosinophils
Eosinophils are granulocytic leukocytes accounting for approximately 1–4% of circulating white blood cells under physiological conditions. They are distinguished morphologically by a bilobed nucleus and large cytoplasmic granules that stain intensely with eosin due to their high content of cationic proteins. Major granule constituents include major basic protein (MBP), eosinophil peroxidase (EPO), eosinophil-derived neurotoxin (EDN), and eosinophil cationic protein (ECP), all of which possess cytotoxic and immunomodulatory properties. Eosinophils play a pivotal role in host defense against helminthic and other multicellular parasitic infections. Their activation is typically driven by type 2 immune responses characterized by cytokines such as IL-5, IL-4, and IL-13, predominantly produced by Th2 lymphocytes and innate lymphoid cells. IL-5 is particularly critical for eosinophil differentiation, activation, and survival. Eosinophils express surface receptors for IgE and IgG (Fc receptors), enabling antibody-dependent cellular cytotoxicity (ADCC). When antibodies coat a parasite, eosinophils bind via Fc receptors and release granule contents onto the target surface, leading to membrane disruption and parasite damage.
In addition to their antiparasitic functions, eosinophils contribute to allergic and atopic disorders, including asthma, allergic rhinitis, and atopic dermatitis. In these contexts, inappropriate or excessive eosinophil activation leads to tissue inflammation and remodeling. Eosinophils also secrete cytokines, growth factors, and lipid mediators (e.g., leukotrienes) that influence vascular permeability, mucus production, and smooth muscle contraction. Eosinophils have broader immunoregulatory roles, including participation in antiviral responses, modulation of adaptive immunity, and maintenance of tissue homeostasis, particularly in mucosal sites. However, sustained eosinophilia can result in organ damage, as observed in hypereosinophilic syndromes. Overall, eosinophils are specialized granulocytes integrating antiparasitic defense with regulation of type 2 inflammatory responses.
Basophils
Basophil are the least abundant circulating granulocytes, typically constituting less than 1% of peripheral blood leukocytes. They possess a lobulated nucleus often obscured by large, basophilic cytoplasmic granules containing histamine, heparin, proteoglycans, and various inflammatory mediators. Although morphologically similar to mast cells, basophils circulate in the bloodstream rather than residing in tissues. Basophils are key effector cells in immediate hypersensitivity reactions and allergic inflammation. They express high-affinity IgE receptors (FcεRI) on their surface. Upon cross-linking of receptor-bound IgE by specific antigen, basophils undergo rapid degranulation, releasing histamine and other mediators that increase vascular permeability, promote vasodilation, and induce smooth muscle contraction. These effects underlie the clinical manifestations of allergic reactions, including urticaria, bronchoconstriction, and, in severe cases, anaphylaxis.
Beyond mediator release, basophils produce cytokines such as IL-4 and IL-13, which promote Th2 polarization and IgE class switching in B cells. Through these functions, basophils contribute to the amplification and maintenance of type 2 immune responses. They also participate in defense against helminths, complementing eosinophil activity by facilitating IgE-mediated immunity. Although historically considered redundant, basophils are now recognized as immunoregulatory cells capable of influencing both innate and adaptive immunity. Abnormal basophil activation is implicated in allergic disorders, chronic urticaria, and certain myeloproliferative diseases. Despite their scarcity, basophils exert disproportionate effects on inflammatory cascades through rapid mediator release and cytokine production, underscoring their significance in hypersensitivity and immune regulation.
RETICULOENDOTHELIAL SYSTEM (RES)
The reticuloendothelial system (RES), more precisely termed the mononuclear phagocyte system (MPS), comprises a distributed network of phagocytic cells that originate in the bone marrow and differentiate into circulating monocytes and tissue-resident macrophages. These cells are strategically positioned in highly vascularized organs where they continuously survey the bloodstream and tissues for foreign antigens, senescent cells, immune complexes, and invading microorganisms. Functionally, the RES is a central component of innate immunity and plays a critical role in maintaining homeostasis. Primary anatomical sites of the RES include the bone marrow, liver, spleen, and lungs. In the liver, specialized macrophages known as Kupffer cells line the hepatic sinusoids and efficiently clear bacteria, endotoxins, and particulate matter from portal and systemic circulation. The spleen contains red pulp macrophages that filter blood-borne pathogens and remove aged erythrocytes, while white pulp regions coordinate antigen presentation and adaptive immune activation. In the lungs, alveolar macrophages provide frontline defense by phagocytosing inhaled microbes and particulates. The bone marrow not only generates monocytes but also contains resident macrophages that regulate hematopoiesis and eliminate cellular debris.
Mechanistically, RES macrophages recognize pathogens through pattern recognition receptors (PRRs) such as Toll-like receptors (TLRs), scavenger receptors, and complement receptors. Upon recognition, microbes are internalized via phagocytosis and destroyed within phagolysosomes through oxidative and enzymatic mechanisms, including the production of reactive oxygen species and lysosomal hydrolases. In addition to direct microbial killing, macrophages process and present antigens through major histocompatibility complex (MHC) molecules, thereby linking innate and adaptive immune responses. The filtering function of the RES is particularly important in preventing systemic dissemination of pathogens. By continuously “sieving” circulating blood, macrophages reduce microbial load before organisms can establish bacteremia or progress to sepsis. This clearance capacity is especially critical in the portal circulation, where microbes translocating from the gastrointestinal tract are intercepted by hepatic macrophages. Impairment of RES function – whether due to immunosuppression, liver dysfunction, splenectomy, or overwhelming microbial burden can significantly increase susceptibility to systemic infections. The reticuloendothelial system functions as a distributed immunological filtration network. Through coordinated phagocytosis, antigen presentation, and immune regulation, it limits microbial spread, supports host defense, and preserves circulatory sterility.
MAST CELLS
Mast cells are granulated immune cells derived from hematopoietic stem cells in the bone marrow and play a central role in initiating rapid inflammatory responses. After differentiation, mast cell precursors migrate from the bloodstream into peripheral tissues, where they complete maturation. They are strategically located in connective tissues, particularly near blood vessels, nerves, and epithelial surfaces, positioning them to act as early responders to infection, injury, and immunological challenges. Although mast cells share morphological and functional similarities with basophils, they are distinct cell types with different developmental lineages and tissue distributions. Basophils are circulating white blood cells characterized by cytoplasmic granules containing inflammatory mediators such as histamine and heparin, an anticoagulant. Unlike mast cells, basophils remain in the bloodstream under normal physiological conditions and are recruited to tissues during inflammatory or allergic responses. Both mast cells and basophils contribute to hypersensitivity reactions and host defense mechanisms; however, mast cells primarily function as tissue-resident sentinel cells, whereas basophils serve as circulating effector cells.
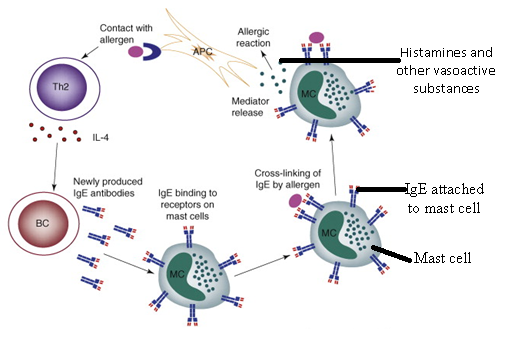
Mast cells respond to diverse external and internal stimuli, including microbial pathogens, physical injury, allergens, and complement activation. Upon activation, they rapidly release a wide array of preformed and newly synthesized pro-inflammatory mediators. Preformed mediators stored in cytoplasmic granules include histamine, proteases, heparin, and various cytokines. Newly synthesized mediators include prostaglandins, leukotrienes, and additional cytokines that amplify and sustain inflammatory responses. These mediators promote vasodilation, increase vascular permeability, stimulate smooth muscle contraction, and facilitate the recruitment of phagocytic cells such as neutrophils and macrophages to sites of tissue damage or infection. A key feature of mast cell activation is the expression of high-affinity receptors for the Fc region of immunoglobulin E (IgE) on their surface. When IgE antibodies bind to these Fc receptors and subsequently cross-link upon encountering a specific antigen such as during exposure to allergens or microbial antigens – it triggers intracellular signaling pathways that lead to mast cell degranulation. Degranulation refers to the rapid exocytosis of granule contents into the extracellular environment. This process releases vasoactive substances and inflammatory mediators that initiate and propagate immediate hypersensitivity reactions.
The mediators released during degranulation include histamine, which increases vascular permeability and induces vasodilation; heparin, which exhibits anticoagulant properties; leukotrienes, which contribute to bronchoconstriction and prolonged inflammation; and prostaglandins, which mediate pain, fever, and additional vascular changes. Collectively, these substances enhance immune cell trafficking and strengthen local immune defenses but may also contribute to pathological inflammation when dysregulated. Mast cells are tissue-resident immune cells that play a critical role in rapid inflammatory and allergic responses. While they share structural similarities with basophils, their localization, activation dynamics, and functional specialization distinguish them within the immune system. Through IgE-mediated activation and degranulation, mast cells release potent mediators that regulate vascular responses, recruit immune cells, and contribute to both protective immunity and hypersensitivity reactions.
DENDRITIC CELLS
Dendritic cells are a specialized population of leukocytes distributed in peripheral and lymphoid tissues, including the skin, thymus, spleen, and lymph nodes. They function primarily as professional antigen-presenting cells (APCs) and play a central role in initiating and regulating adaptive immune responses. By capturing, processing, and presenting antigenic material to T lymphocytes, dendritic cells provide a critical link between innate and adaptive immunity. Morphologically, dendritic cells are characterized by their irregular shape and the presence of long membranous cytoplasmic extensions that resemble neuronal dendrites. These projections increase their surface area and enhance their ability to sample antigens from the surrounding microenvironment. Although they are often described as motile cells, they are typically non-phagocytic under steady-state conditions compared with macrophages. Their cytoplasm contains abundant mitochondria to support metabolic activity, and they possess a distinct nucleus that contributes to their functional specialization.
A defining feature of dendritic cells is the constitutive or inducible expression of major histocompatibility complex (MHC) class II molecules. Through these molecules, they process exogenous antigens and present peptide fragments to CD4⁺ T-helper cells. This interaction, together with the expression of co-stimulatory molecules and cytokine secretion, determines the nature of the subsequent T-cell response, including activation, differentiation, or tolerance induction. Because of this capability, dendritic cells are regarded as the most potent antigen-presenting cells in the immune system. Several subsets of dendritic cells are identified based on tissue localization and functional characteristics. Langerhans cells reside in the epidermis and represent a specialized dendritic population involved in cutaneous immune surveillance. Interdigitating dendritic cells are commonly found within lymph nodes and other secondary lymphoid organs, where they present antigens to naïve T cells during immune activation. Additional subsets have also been described in blood and tissues, reflecting the heterogeneity of this cell lineage. Dendritic cells are essential regulators of immune homeostasis and defense, coordinating antigen detection with the activation of adaptive immune mechanisms.
REFERENCES
Abbas A.K, Lichtman A.H and Pillai S (2010). Cellular and Molecular Immunology. Sixth edition. Saunders Elsevier Inc, USA.
Actor J (2014). Introductory Immunology. First edition. Academic Press, USA.
Alberts B, Bray D, Johnson A, Lewis J, Raff M, Roberts K and Walter P (1998). Essential Cell Biology: An Introduction to the Molecular Biology of the Cell. Third edition. Garland Publishing Inc., New York.
Bach F and Sachs D (1987). Transplantation immunology. N. Engl. J. Med. 317(8):402-409.
Barrett J.T (1998). Microbiology and Immunology Concepts. Philadelphia, PA: Lippincott-Raven Publishers. USA.
Jaypal V (2007). Fundamentals of Medical Immunology. First edition. Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India.
John T.J and Samuel R (2000). Herd Immunity and Herd Effect: New Insights and Definitions. European Journal of Epidemiology, 16:601-606.
Levinson W (2010). Review of Medical Microbiology and Immunology. Twelfth edition. The McGraw-Hill Companies, USA.
Roitt I, Brostoff J and Male D (2001). Immunology. Sixth edition. Harcourt Publishers Limited, Spain.
Zon LI (1995). Developmental biology of hematopoiesis. Blood, 86(8): 2876–91.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.