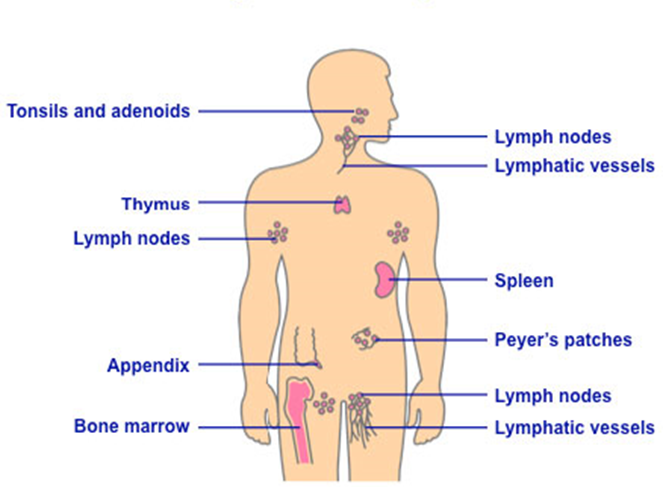
The organs of the immune system are classically categorized into two principal groups: primary (central) lymphoid organs and secondary (peripheral) lymphoid organs. This structural and functional organization underpins the generation, maturation, and activation of adaptive immune responses. Together, these organs coordinate the recognition of antigens, the clonal expansion of lymphocytes, and the establishment of immunological memory.
Primary lymphoid organs are responsible for lymphopoiesis and the antigen-independent maturation of lymphocytes. In mammals, these include the bone marrow and the thymus. The bone marrow serves as the site of hematopoiesis, where multipotent hematopoietic stem cells differentiate into all blood cell lineages, including B lymphocytes, T lymphocyte precursors, natural killer (NK) cells, and other immune effector cells. B cells undergo maturation and central tolerance induction within the bone marrow. The thymus provides a specialized microenvironment for the maturation and selection of T lymphocytes. Through positive and negative selection processes, the thymus ensures that emerging T cells are both self-major histocompatibility complex (MHC) restricted and self-tolerant, thereby minimizing the risk of autoimmunity.
Secondary lymphoid organs are anatomically positioned to facilitate antigen capture and the initiation of adaptive immune responses. These include the spleen, lymph nodes, tonsils, Peyer’s patches, and other components of mucosa-associated lymphoid tissue (MALT). Lymph nodes filter lymphatic fluid draining from peripheral tissues, enabling antigen-presenting cells to interact with naïve T and B cells in highly organized microanatomical compartments. The spleen performs a similar function for blood-borne antigens, while also contributing to erythrocyte turnover and immune surveillance. MALT, which encompasses structures in the gastrointestinal, respiratory, and genitourinary mucosae, provides localized immune protection at major portals of pathogen entry. Peyer’s patches, located in the small intestine, are specialized for sampling luminal antigens and generating mucosal immune responses, particularly IgA-mediated immunity.
The liver, particularly in early life, contributes to hematopoiesis and hosts a substantial population of innate immune cells, including Kupffer cells, that participate in systemic immune regulation. These organs form an integrated and highly specialized network. Their coordinated functions distinguish the immune system from other physiological systems, enabling precise discrimination between self and non-self and mounting robust, antigen-specific defenses against invading pathogens.
The organs of the immune system provide the structural foundation for adaptive immunity. Primary lymphoid organs bone marrow and thymus generate and educate lymphocytes, establishing self-tolerance and receptor diversity. Secondary lymphoid organs such as the spleen and tonsils serve as platforms for antigen encounter and immune activation. Together, these specialized anatomical sites ensure that immune responses are precise, regulated, and capable of protecting the host against a vast array of pathogens while minimizing damage to self-tissues.
Understanding the Organs of the Immune System
A rigorous understanding of the organs of the immune system is fundamental to immunology because immune competence depends not only on cellular and molecular mechanisms but also on the anatomical microenvironments in which immune cells develop, differentiate, and interact. The immune system is structurally organized into primary (central) and secondary (peripheral) lymphoid organs, each fulfilling distinct but coordinated roles in host defense.
Primary (Central) Lymphoid Organs
Primary lymphoid organs are the sites of lymphocyte development, differentiation, and central tolerance. These organs generate immunocompetent B and T lymphocytes capable of participating in adaptive immune responses while maintaining self-tolerance.
The two primary lymphoid organs are:
- Bone marrow
- Thymus
Bone Marrow
The bone marrow is the principal site of hematopoiesis, the process by which all blood cells originate from multipotent hematopoietic stem cells. Within this microenvironment:
- B lymphocytes undergo development and maturation.
- T lymphocyte precursors are generated before migrating to the thymus.
- Myeloid cells (e.g., neutrophils, monocytes, dendritic cells) are produced.
During B-cell development in the bone marrow, immunoglobulin gene rearrangement (V(D)J recombination) generates unique antigen receptor specificities. Immature B cells expressing self-reactive receptors are eliminated or edited through central tolerance mechanisms, reducing the risk of autoimmunity. Thus, the bone marrow serves both as a generative site and as a quality-control checkpoint for emerging lymphocytes.
Thymus
The Thymus is a bilobed primary lymphoid organ situated in the anterior mediastinum, immediately superior to the heart and posterior to the sternum. It is the central organ responsible for the maturation and functional education of T lymphocytes. Although T-cell precursors originate in the bone marrow, they migrate via the bloodstream to the thymus, where they differentiate into immunocompetent yet self-tolerant T cells.
Within the thymic microenvironment composed of cortical and medullary epithelial cells, dendritic cells, and macrophages developing thymocytes undergo a tightly regulated maturation program. This process includes:
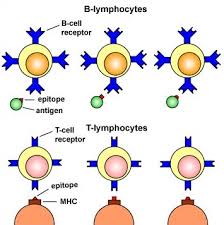
- T-cell receptor (TCR) gene rearrangement – Somatic recombination of TCR gene segments generates a highly diverse repertoire of antigen receptors, ensuring broad recognition capacity against potential pathogens.
- Positive selection – Occurring primarily in the thymic cortex, this step preserves thymocytes capable of recognizing self–major histocompatibility complex (MHC) molecules with appropriate affinity. Cells that fail to recognize self-MHC undergo apoptosis.
- Negative selection – Taking place mainly in the thymic medulla, this process eliminates thymocytes that bind strongly to self-antigens presented by MHC molecules, thereby establishing central tolerance and reducing the risk of autoimmunity.
Only a small fraction of thymocytes survive these selection checkpoints. The resulting mature CD4⁺ or CD8⁺ T cells exit the thymus and enter peripheral circulation as naïve but immunocompetent lymphocytes capable of mounting adaptive immune responses. This dual selection process ensures that mature T cells are both MHC-restricted and self-tolerant. The thymus is most active during childhood and undergoes physiological involution with age, although functional T-cell production persists throughout life.
Development of Immunocompetence
Primary lymphoid organs principally the bone marrow and thymus are the anatomical sites where lymphocytes acquire the defining properties of adaptive immunity. Within these controlled microenvironments, developing B and T cells undergo genetic rearrangement, selection, and functional programming that collectively establish immunocompetence. The key attributes acquired during this developmental phase include antigen specificity, clonal diversity, immunological memory potential, and self–non-self-discrimination. Primary lymphoid organs are where lymphocytes acquire the defining features of adaptive immunity. These defining features of the adaptive immunity are:
- Antigen specificity
- Clonal diversity
- Immunological memory potential
- Self–non-self-discrimination
Antigen specificity arises through somatic recombination of antigen receptor genes. In B cells, immunoglobulin (Ig) gene segments variable (V), diversity (D), and joining (J) rearrange to produce a unique B-cell receptor (BCR). In T cells, a comparable V(D)J recombination process generates distinct T-cell receptors (TCRs). This genetic reorganization ensures that each lymphocyte expresses a single receptor specificity capable of recognizing a particular epitope. Consequently, adaptive immunity operates with remarkable precision, targeting defined molecular structures rather than broad pathogen classes.
Clonal diversity refers to the vast repertoire of lymphocytes generated through combinatorial gene rearrangement, junctional diversity, and pairing of receptor chains. This process produces millions of distinct antigen receptor configurations, enabling the immune system to anticipate and respond to an extensive array of potential pathogens, including those not previously encountered.
Immunological memory potential is programmed during lymphocyte maturation, although memory differentiation occurs after antigen exposure in secondary lymphoid organs. Mature naïve lymphocytes are intrinsically capable of undergoing clonal expansion and differentiating into long-lived memory cells following activation. This capacity underpins the accelerated and amplified responses observed during secondary exposures to the same antigen.
Self–non-self-discrimination is established through central tolerance mechanisms. In the bone marrow and thymus, lymphocytes that strongly recognize self-antigens are eliminated or functionally inactivated through negative selection. This process minimizes autoreactivity and is critical for preventing autoimmune disease while preserving protective immunity.
It is critical to clarify that lymphocytes do not become committed to a specific antigen within primary lymphoid organs. Rather, they acquire unique antigen receptor specificity through gene rearrangement before encountering their cognate antigen. True antigen-driven activation and clonal expansion occur later in secondary lymphoid organs. At the end of maturation, naïve but immunocompetent B and T lymphocytes exit the primary organs and circulate via blood and lymph to peripheral tissues.
Secondary (Peripheral) Lymphoid Organs
Secondary lymphoid organs are the sites where mature lymphocytes encounter antigens and initiate adaptive immune responses. These organs are strategically positioned to monitor tissues and body fluids for invading pathogens.
Major secondary lymphoid organs include:
- Spleen
- Tonsils
- Lymph nodes (distributed throughout the body)
- Mucosa-associated lymphoid tissue (MALT)
Tonsils and Adenoids
The tonsils are aggregates of lymphoid tissue forming part of Waldeyer’s ring in the pharyngeal region. Tonsils are lymphoid aggregates located at the entrance of the respiratory and gastrointestinal tracts. They sample inhaled and ingested antigens, facilitating early immune responses. By promoting IgA production and lymphocyte activation, they provide frontline defense against pathogens entering through the mouth and nasal passages. They provide immunological surveillance at the entrance of the respiratory and gastrointestinal tracts.
The major components include:
- Palatine tonsils – located at the lateral walls of the oropharynx
- Adenoids – situated in the roof of the nasopharynx
- Lingual tonsils – located at the base of the tongue
These structures sample inhaled and ingested antigens. Specialized epithelial cells facilitate antigen uptake and presentation to underlying lymphocytes. This promotes local IgA production and the generation of adaptive responses against pathogens entering via the mouth or nasal passages.
Because they are frequently exposed to environmental antigens, tonsils are common sites of inflammation, particularly in children.
Spleen
The spleen is the largest secondary lymphoid organ and is located in the left upper quadrant of the abdominal cavity. Unlike lymph nodes, which filter lymphatic fluid, the spleen filters blood. The spleen is the largest secondary lymphoid organ and specializes in filtering blood rather than lymph. It removes senescent erythrocytes and traps blood-borne pathogens. Its white pulp contains organized B- and T-cell zones where adaptive immune responses are initiated against circulating antigens.
The functions of the spleen include:
- Removal of senescent erythrocytes
- Clearance of blood-borne pathogens
- Immune surveillance of circulating antigens
The spleen contains two major compartments:
- White pulp – lymphoid tissue organized around central arteries, where B and T cells initiate immune responses.
- Red pulp – responsible for erythrocyte filtration and iron recycling.
The spleen is particularly important in defense against encapsulated bacteria circulating in the bloodstream. Individuals without a functional spleen are at increased risk of overwhelming infections and require appropriate vaccination and prophylactic strategies.
Lymph nodes
Lymph nodes are strategically distributed along lymphatic vessels throughout the body. They filter lymphatic fluid, trapping antigens drained from peripheral tissues. Within distinct cortical and paracortical regions, B and T lymphocytes interact with antigen-presenting cells to initiate adaptive immune responses.
Mucosa-associated lymphoid tissue (MALT)
MALT comprises diffuse and organized lymphoid tissues located along mucosal surfaces, including the gastrointestinal, respiratory, and genitourinary tracts. It provides localized immune surveillance at sites of constant environmental exposure, promoting IgA-mediated protection and maintaining immune tolerance to commensal microorganisms.
Functional Integration of Immune Organs
The immune system operates as a coordinated anatomical and cellular network:
- Bone marrow generates lymphocytes.
- The thymus educates T cells.
- Secondary organs concentrate antigens and lymphocytes to facilitate efficient immune activation.
- Activated lymphocytes migrate to sites of infection to exert effector functions.
This compartmentalization enhances both specificity and efficiency. Primary organs ensure diversity and tolerance; secondary organs enable antigen recognition, clonal expansion, and effector differentiation.
Organization and Functional Roles of Key Immune Organs
The organs of the immune system form an integrated anatomical network that supports immune cell development, antigen surveillance, and the initiation of adaptive immune responses. Each organ provides a specialized microenvironment that ensures efficient coordination between innate and adaptive immunity.
The Bone marrow is the principal site of hematopoiesis and B-cell maturation. Within its stromal microenvironment, hematopoietic stem cells differentiate into multiple blood cell lineages, including red blood cells (erythrocytes), white blood cells (leukocytes), and platelets. Critically, B lymphocytes undergo immunoglobulin gene rearrangement and central tolerance screening in the bone marrow. Self-reactive immature B cells are either eliminated, edited, or rendered anergic, ensuring that mature B cells released into circulation are immunocompetent yet self-tolerant.
Lymph nodes are distributed throughout the body along lymphatic vessels, particularly at vascular junctions. They function as biological filtration units for lymphatic fluid drained from peripheral tissues. Structurally organized into cortical (B-cell zones), paracortical (T-cell zones), and medullary regions, lymph nodes provide an optimized microenvironment for antigen capture, processing, and presentation. Dendritic cells transport antigens from tissues to lymph nodes, where they present peptide fragments to naïve T lymphocytes, initiating clonal expansion and differentiation.
The Spleen, located in the left upper abdominal cavity beneath the pancreas, specializes in filtering blood rather than lymph. Its red pulp removes aged erythrocytes, while its white pulp contains lymphoid follicles and periarteriolar lymphoid sheaths where adaptive immune responses to blood-borne pathogens are initiated. The spleen is particularly important in protection against encapsulated bacteria circulating systemically.
Within the gastrointestinal tract, Peyer’s patches represent organized aggregates of lymphoid tissue embedded in the intestinal mucosa. They monitor luminal antigens and coordinate mucosal immune responses, particularly IgA production. Similar lymphoid functions are observed in the appendix, which also contains substantial immune tissue.
The Lymphatic vessels serve as conduits that transport interstitial fluid, immune cells, and antigens from tissues to regional lymph nodes. By distributing antigenic material to secondary lymphoid organs, the lymphatic system ensures that localized infections can trigger specific, systemic adaptive immune responses.
e=”background-color: #fee894″>In birds, B cell development occurs in the Bursa of Fabricius. After their initial development in the bone marrow, lymphocytes leave to complete their maturation in the peripheral lymphoid organs (i.e., the secondary lymphoid organs).
Immunocompetence, Lymphocyte Maturation, and Clonal Selection in the Immune System
B-cell development and maturation occur within the Bone marrow, whereas T-cell maturation takes place in the Thymus. A lymphocyte attains immunocompetence only after completing its maturation program within these primary lymphoid organs. Immunocompetent B and T cells are those that have successfully undergone genetic receptor rearrangement and selection processes, enabling them to recognize specific antigens and mount effective immune responses against pathogens in animals and mammals.
Although maturation occurs prior to antigen encounter, functional competence is confirmed through controlled interactions with self- and non-self-molecules. Lymphocytes become fully operational after passing stringent selection checkpoints that assess receptor functionality and self-tolerance. At this stage, they circulate as naïve but immunologically competent cells, capable of responding rapidly upon exposure to their cognate antigen. True activation and clonal expansion occur after antigen recognition in secondary lymphoid organs, where antigen-presenting cells provide the necessary stimulatory signals.
A central mechanism underlying immunocompetence is clonal selection. During this process, lymphocytes expressing receptors with specificity for foreign antigens are positively selected for survival and proliferation. In contrast, cells that bind strongly to self-antigens undergo negative selection and are eliminated or rendered inactive. This mechanism ensures that the immune system mounts responses against invading pathogens while minimizing the risk of autoimmune reactions. Through clonal selection, antigen-specific lymphocyte clones expand to form effector and memory populations.
The bone marrow serves as the origin of all hematopoietic lineages, generating not only lymphocytes but also erythrocytes, platelets, and various myeloid cells. Its role as a hematopoietic and immunological foundation highlights its importance in maintaining both immune defense and overall physiological homeostasis.
Bone Marrow: Central Site of Hematopoiesis and Immune Cell Development
The Bone marrow is a yellowish, soft, highly vascularized tissue located within the medullary cavities of major bones, particularly long bones, the pelvis, ribs, sternum, and vertebrae. It represents the principal site of hematopoiesis in adults and serves as the foundational origin of all cellular components of the immune and blood systems. Through tightly regulated stem cell differentiation processes, hematopoietic stem cells (HSCs) give rise to diverse immune lineages as well as other essential blood elements, ensuring systemic homeostasis and immune competence.
During hematopoiesis, HSCs differentiate into common lymphoid and myeloid progenitors. B lymphocytes mature directly within the bone marrow, whereas T-cell precursors originate there but migrate to the thymus for further maturation. In addition to lymphocytes, the bone marrow generates multiple immune and non-immune cell types, including mast cells, dendritic cell precursors, natural killer (NK) cells, erythrocytes (red blood cells), platelets, and granulocytes such as neutrophils, eosinophils, and basophils. This broad cellular output highlights its dual role in both immunity and general physiological maintenance.
After maturation in primary lymphoid organs, lymphocytes exit into the peripheral circulation and subsequently encounter antigens within secondary lymphoid organs. B cells differentiate into antibody-secreting plasma cells and long-lived memory B cells upon activation. Plasma cells mediate humoral immunity through targeted antibody production, whereas memory B cells provide rapid and amplified responses upon re-exposure to the same antigen. Similarly, T cells differentiate into effector T cells such as helper T cells and cytotoxic T lymphocytes and memory T cells that sustain long-term cell-mediated immunity. These differentiated populations interact with antigens within lymph nodes, spleen, and mucosal lymphoid tissues to mount coordinated immune responses.
During early development, B-cell maturation does not exclusively occur in adult bone marrow. In neonates and fetuses, B-cell development initially takes place in the fetal liver, fetal bone marrow, and yolk sac before transitioning fully to the adult bone marrow after birth. In avian species, B-cell maturation occurs in the bursa of Fabricius, a specialized lymphoid organ that fulfills the functional equivalent of mammalian bone marrow for B-cell education.
A critical feature of bone marrow lymphopoiesis is clonal selection and central tolerance. B cells expressing receptors with high affinity for self-antigens are typically eliminated, edited through receptor modification, or rendered functionally inactive. This negative selection process prevents the emergence of autoreactive clones that could initiate autoimmune disease. A similar principle governs T-cell maturation in the thymus, where developing T cells that strongly recognize self-antigens presented in the context of major histocompatibility complex (MHC) molecules are deleted during negative selection. Together, these mechanisms ensure that only lymphocytes capable of recognizing non-self-antigens survive and proliferate.
Through this rigorous selection process, lymphocytes are “educated” to target foreign pathogens while maintaining tolerance to self-components. Only lymphocyte populations with appropriate antigenic specificity are permitted to expand and differentiate into effector and memory cells. Beyond immune cell generation, the bone marrow remains essential for producing erythrocytes, which transport oxygen; platelets, which mediate blood clotting; and various myeloid cells that contribute to innate immunity and tissue repair. Its multifunctional role underscores its importance as both a hematopoietic organ and a central regulator of immune system development and systemic physiological balance.
Thymus Function and the Role of Secondary Lymphoid Organs in Adaptive Immunity
The Thymus is a bilobed lymphoid organ situated in the anterior mediastinum, positioned superior to the heart and posterior to the sternum. It serves as the principal site for the maturation and functional education of immature T-cell precursors that originate from the bone marrow. Although these progenitor cells arise in the bone marrow, they migrate to the thymus to complete their differentiation into immunocompetent T lymphocytes.
Structurally, the thymus is organized into two major compartments: the outer cortex and the inner medulla. Both regions contain developing T cells, known as thymocytes, at distinct stages of maturation. The cortical region is densely populated with immature thymocytes undergoing T-cell receptor (TCR) gene rearrangement and positive selection. During positive selection, thymocytes that are capable of recognizing self–major histocompatibility complex (MHC) molecules with appropriate affinity are retained, while those unable to recognize MHC molecules undergo apoptosis.
In the medullary region, thymocytes undergo negative selection. This process eliminates T cells that bind strongly to self-antigens presented by antigen-presenting cells. Negative selection is essential for establishing central tolerance and preventing the release of autoreactive T cells into the peripheral circulation. Through these tightly regulated selection mechanisms, the thymus ensures the continuous production of a diverse but self-tolerant pool of immunocompetent T lymphocytes capable of responding to foreign antigens presented by MHC molecules.
After maturation in primary lymphoid organs, lymphocytes migrate to secondary lymphoid organs, where antigen encounter and immune activation occur. Secondary lymphoid organs are specialized anatomical sites that support lymphocyte activation, clonal expansion, and differentiation into effector and memory cells. Unlike primary organs, which focus on development and selection, secondary organs function as immunological interaction hubs.
Examples of secondary lymphoid organs include lymph nodes, the spleen, and mucosal lymphoid tissues. These structures are strategically positioned along lymphatic vessels and blood circulation pathways, enabling efficient antigen sampling from peripheral tissues. Within these microenvironments, antigen-presenting cells display processed antigenic peptides to naïve B and T cells, triggering activation and proliferation.
B cells activated within secondary lymphoid organs differentiate into antibody-secreting plasma cells and long-lived memory B cells. Plasma cells mediate antibody-mediated (humoral) immunity by producing antigen-specific immunoglobulins that neutralize pathogens and facilitate their clearance. Memory B cells persist after infection and enable rapid, amplified responses upon re-exposure to the same antigen.
Similarly, activated T cells differentiate into effector T cells and memory T cells. Effector T cells including helper T cells and cytotoxic T lymphocytes mediate cell-mediated immunity (CMI) by coordinating immune responses or directly eliminating infected or abnormal cells. Memory T cells provide long-term immune surveillance and ensure enhanced responsiveness during secondary infections. Secondary lymphoid organs provide the structural and cellular environment required for antigen recognition and adaptive immune activation. By integrating lymphocyte trafficking, antigen presentation, and cellular interaction, these organs bridge lymphocyte development in primary tissues with functional immune defense in peripheral sites.
Structure and Immunological Function of the Lymphatic System and Secondary Lymphoid Organs
The lymphatic system is an extensive network of vessels, tissues, and organs responsible for maintaining fluid balance and facilitating immune surveillance. It consists of thin-walled lymphatic vessels that collect interstitial fluid from peripheral tissues and transport it as lymph – a pale biological fluid containing water, proteins, cellular debris, and immune cells such as white blood cells (WBCs) back into the bloodstream. In addition to its role in fluid homeostasis, the lymphatic system functions as a critical conduit for antigen transport and immune cell trafficking.
As lymph drains from tissues, it carries antigens, pathogens, and antigen-presenting cells toward secondary lymphoid organs, where adaptive immune responses are initiated. The interaction between lymphocytes and antigens within these specialized microenvironments leads to clonal expansion and differentiation into effector cells with defined antigen specificity. This process ensures that immune responses are targeted and amplified against invading pathogens. Major secondary lymphoid organs include lymph nodes, the spleen, and mucosa-associated lymphoid tissue (MALT), all of which coordinate antigen detection and immune activation.
Secondary lymphoid organs function as immunological hubs where antigen presentation, lymphocyte activation, and immune cell communication occur. Among these structures, the lymph nodes and spleen are considered the most important due to their strategic anatomical distribution and central role in immune regulation. They provide organized microenvironments that facilitate interactions between antigen-presenting cells and naïve or memory lymphocytes, enabling efficient initiation of adaptive immunity.
Lymph nodes are encapsulated, bean-shaped structures distributed throughout the body, particularly along lymphatic vessel junctions. They form part of a highly organized immune network composed of B lymphocytes, T lymphocytes, macrophages, and dendritic cells arranged in distinct functional regions. The cortex contains B-cell follicles, the paracortex is enriched with T cells and antigen-presenting cells, and the medulla supports antibody-producing plasma cells. Lymph nodes act as biological filters that trap antigens from lymph fluid before it re-enters circulation. Their rich supply of afferent and efferent lymphatic vessels ensures continuous sampling of peripheral tissue-derived antigens and efficient immune surveillance.
Within lymph nodes, dendritic cells migrate from infected tissues carrying processed antigens and present them to immunocompetent lymphocytes. This antigen presentation triggers clonal expansion and differentiation into effector and memory cells, forming the basis of adaptive immune protection. Activated B cells may differentiate into plasma cells that secrete antigen-specific antibodies, while activated T cells develop into helper or cytotoxic effector cells that eliminate infected cells.
Beyond lymph nodes, the Spleen filters blood-borne antigens and plays a vital role in immune responses against systemic infections. It contains white pulp for immune activation and red pulp for blood filtration. Mucosa-associated lymphoid tissues (MALT) further extend immune surveillance to mucosal surfaces such as the gastrointestinal and respiratory tracts, providing localized protection against environmental pathogens. The lymphatic system and its associated secondary lymphoid organs ensure continuous antigen monitoring, lymphocyte activation, and coordinated immune defense throughout the body.
Note: while lymph nodes trap antigens from lymph, the spleen traps antigens in the blood. The mucosal-associated lymphoid tissues (MALT) are secondary lymphoid organs lining mucous membranes of the GIT, urogenital tract, respiratory tract, and other mucous membrane surfaces in the body, defending these surfaces against pathogenic microorganisms or antigens.
The germinal center of secondary follicles is densely populated with B lymphocytes. Lymph nodes mount immune responses to antigenic molecules in lymph fluids and are mainly located at the junctions of lymphatic vessels throughout the body. Phagocytic cells and dendritic cells within the lymph nodes trap particulate antigens or pathogens entering via lymph; lymphocytes then respond to mount an immune response against trapped antigens.
The cortex, paracortex, and medulla are the three main sections of the lymph node (Figure 2). The cortex is the outermost part, rich in macrophages, lymphocytes (particularly B cells), and dendritic cells. The paracortex underlies the cortex and is rich in T lymphocytes, with dendritic cells also found in this area. The medulla is the innermost part of the lymph node, sparsely populated with antibody-secreting plasma cells and other cells of lymphoid origin.
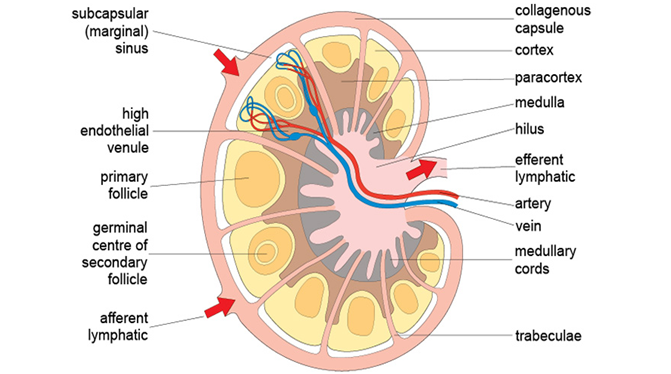
Lymph nodes trap foreign bodies or antigens before returning lymph fluids to systemic circulation. Activated lymphocytes in lymph nodes proliferate and differentiate into numerous effector cells following antigen invasion. After antigen interaction with macrophages or dendritic cells in lymph nodes, the antigen is processed and presented to immunocompetent B and T cells for instigating appropriate immune responses against pathogens.
Lymph fluids containing antigens or pathogens enter lymph nodes via the afferent lymph duct, while the efferent lymphatics are the passageways through which immunoglobulins and lymphocytes leave the lymph nodes to the bloodstream (Figure 2). The spleen is an oval-shaped organ located in the upper left abdominal cavity; it is the site where antigens or pathogens are trapped from blood circulation, and outworn RBCs are destroyed.
The spleen, unlike lymph nodes, primarily filters blood that passes through it, trapping antigens and presenting them to lymphocytes in peripheral lymphoid tissues. It also traps antigens from local tissues aside from those carried through blood. Lymphocytes and blood-borne antigens are carried into the spleen via blood vessels (e.g., spleen arteries); foreign bodies are presented to lymphocytes for appropriate immune responses.
Functional Roles of Mucosa-Associated Lymphoid Tissue and Antigen Surveillance
Secondary lymphoid organs differ in the compartments of the body they monitor. Lymph nodes primarily filter and trap antigens transported through lymphatic fluid, whereas the Spleen filters blood and captures circulating pathogens. This division of labor ensures comprehensive immune surveillance across both interstitial fluid and systemic circulation.
Mucosa-associated lymphoid tissue (MALT) represents a specialized group of secondary lymphoid tissues distributed along mucosal surfaces, including the gastrointestinal tract (GIT), respiratory tract, urogenital tract, and other epithelial barriers exposed to the external environment. Because these surfaces are constantly exposed to microorganisms, environmental antigens, and commensal microbes, MALT serves as the first line of adaptive immune defense at mucosal interfaces. Its primary function is to prevent pathogen invasion and colonization while maintaining tolerance to beneficial microbiota.
MALT contains organized lymphoid structures capable of supporting antigen capture, lymphocyte activation, and differentiation. Upon antigen stimulation, activated B cells within these tissues differentiate into plasma cells that produce immunoglobulins—particularly IgA—tailored to neutralize pathogens at mucosal surfaces. These antibodies provide localized protection by preventing microbial adherence and facilitating pathogen clearance without triggering excessive inflammation.
Key anatomical components of MALT include the Peyer’s patches, which are lymphoid aggregates located in the ileum of the small intestine and play a crucial role in sampling intestinal antigens. Another important structure is the Tonsils, located at the base of the tongue and within the pharyngeal region. Tonsils monitor inhaled and ingested antigens and initiate local immune responses when necessary. Together, MALT structures maintain mucosal immunity through continuous antigen surveillance, localized antibody production, and rapid immune activation. This system ensures effective protection at vulnerable entry points while preserving tissue integrity and immune balance.
REFERENCES
Abbas A.K, Lichtman A.H and Pillai S (2010). Cellular and Molecular Immunology. Sixth edition. Saunders Elsevier Inc, USA.
Actor J (2014). Introductory Immunology. First edition. Academic Press, USA.
Alberts B, Bray D, Johnson A, Lewis J, Raff M, Roberts K and Walter P (1998). Essential Cell Biology: An Introduction to the Molecular Biology of the Cell. Third edition. Garland Publishing Inc., New York.
Bach F and Sachs D (1987). Transplantation immunology. N. Engl. J. Med. 317(8):402-409.
Barrett J.T (1998). Microbiology and Immunology Concepts. Philadelphia, PA: Lippincott-Raven Publishers. USA.
Jaypal V (2007). Fundamentals of Medical Immunology. First edition. Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India.
John T.J and Samuel R (2000). Herd Immunity and Herd Effect: New Insights and Definitions. European Journal of Epidemiology, 16:601-606.
Levinson W (2010). Review of Medical Microbiology and Immunology. Twelfth edition. The McGraw-Hill Companies, USA.
Roitt I, Brostoff J and Male D (2001). Immunology. Sixth edition. Harcourt Publishers Limited, Spain.
Zon LI (1995). Developmental biology of hematopoiesis. Blood, 86(8): 2876–91.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.