Malaria in humans is majorly caused by four (4) species of Plasmodium. Plasmodium species are in the Phylum Alveolata, Subphylum Apicomplexa, Class Haematozoa,Order Haemosporida, and Genus Plasmodium. Plasmodium parasite is naturally transmitted to susceptible human hosts from an insect vector called female Anopheles mosquito. The 4 major and well known infectious species of Plasmodium that cause malaria in humans are:
- Plasmodium falciparum
- Plasmodium vivax
- Plasmodium malariae
- Plasmodium ovale
Of these four species, P. falciparum is known to be extremely harmful and most aggressive of them all. P. falciparum it accounts for the main global burden of malaria. The genus Plasmodium is classified as a blood sporozoan. Species of parasites classified as sporozoa (singular: sporozoan)are known to undergo a complex form of life cycle with alternating asexual and sexual reproductive stages that usually occur in two different hosts (a human and an arthropod).
Other examples of blood sporozoan includes: Toxoplasma gondii, Isospora belli, and Cryptosporidium. Plasmodium species can also cause fever-like illnesses in rodents, but the species responsible for malaria in these animals are quite different from those that cause malaria in humans. The rodent malaria parasites are as follows:
- Plasmodium yoelii
- Plasmodium vinckei
- Plasmodium berghei
- Plasmodium chabaudi
- Plasmodium knowlesi is parasitic in monkeys.
Pathogenesis and life cycle of the malaria parasite (Plasmodium)
Inside the human body, the Plasmodium parasite attacks the liver cells which are their first spot of call upon invasion. They penetrate into the liver cells and destroy the cells in which they reside (including the red blood cells). Anaemic conditions (i.e. shortage of blood in the human host) may arise since the Plasmodium parasitedestroyssome liver cells and many erythrocytes (red blood cells).
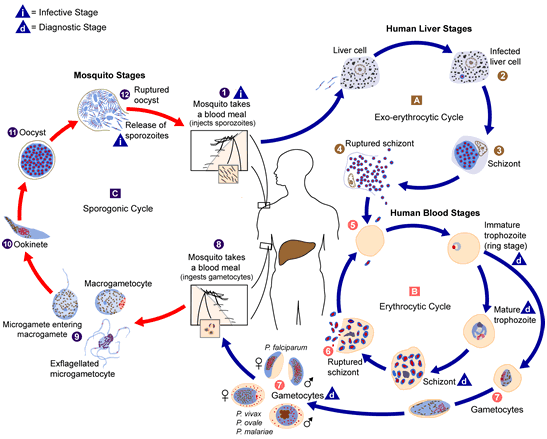
This condition can lead to death following the drastic reduction in oxygen supply, hormonal circulation, and nutrient (food) circulation to cells of the body. The Plasmodium parasite is usually picked and ingested by the insect vector during a blood meal in which it ingests viable plasmodial gametocytes that develops in the insect’s gut (Figure 1). This eventually transforms into the infective forms (sporozoites) that appears in the vector’s salivary gland and is passed on to uninfected human host during the insect’s next blood meal.
After inoculation by the female anopheles mosquito, the Plasmodium parasites mature first in the liver of the human host for a period of about one week before it goes on to complete its multiplication in the red blood cell. This continues until it reaches levels that cause fever (a rise in body temperature) and a wide variety of clinical signs and symptoms in the infected host. The spleen is another vital organ which plays an important role in malaria disease because it is known to confer some level of immunity or protection against malaria to the host.
The spleen usually enlarges during acute malaria as it helps to remove erythrocytes that have been invaded by Plasmodium parasites. Malaria disease causes a high mortality in asplenic individuals (those whose spleen has been surgically removed); and such individuals stand a high risk of P. falciparum infection because there spleen is no longer there to perform its usual flushing and cleansing action.
Pre-erythrocytic stage
Pre-erythrocytic stage which can also be called the exo-erythrocytic cycle is the first stage of Plasmodium parasite development in humans, and it occurs inside the cells of the liver (hepatocytes). It is the stage of the Plasmodium parasite development that occurs in the liver cells of the human host (outside the red blood cells) immediately after the introduction or inoculation of sporozoites (infective stage of Plasmodium parasite) into the human blood stream after the bite of an infected female Anopheles mosquito. In this stage, the Plasmodium parasite only multiplies in the hepatocytes of the human liver.
After a successful blood meal (or bite from a female Anopheles mosquito), sporozoites leaves the salivary gland of the mosquito and find their way to the bloodstream of the human host where they circulate for a little time period before attacking or entering the hepatocytes. The infection of the liver cells (hepatic infection) in humans is usually asymptomatic (i.e. without any fever or cold), and this may last for several days. The sporozoites remain in the hepatocytes for about 10 days for replication and, eventually mature into schizonts.
Upon the rupturing of the schizonts in the hepatocytes, thousands of merozoites are released into the human bloodstream. This marks the beginning of the erythrocytic stage (i.e. the invasion of the red blood cells). It is noteworthy that relapsing fever (or recurrent malaria infection) can also occur in humans after successful treatment, when the dormant stage (hypnozoites) of some Plasmodium species (in particular: sporozoites of P. vivax and P. ovale) persist in the liver; and in turn invade the erythrocytes to cause relapsing fever weeks or years later.
The merozoites do not return again to the liver cells from the bloodstream after its departure or release from hepatocytes. This implies that malaria infection (P. falciparum infection) can naturally terminate without any therapy in a year except the disease ends in the death of the sufferer.
Erythrocytic stage
Erythrocytic stage is the phase of Plasmodium parasite invasion into the red blood cells (RBCs), and which usually marks the clinical manifestation of the malaria disease. It is the stage where the Plasmodium manifests itself in the bloodstream with several nuclear division and replication. The erythrocytic stage usually has two phases: the asexual stage (where schizogony are formed) and sexual stage (where gametocytes are formed).
The asexual stage is marked by the invasion of the erythrocyte by merozoites after their release from the liver cells. The Plasmodium parasite undergo asexual multiplication in the erythrocytes (this is known as erythrocytic schizogony), and the merozoites mature inside the RBCs from a ring to a mature trophozoite. The ring stage trophozoites then undergo asexual division (schizogony) to form schizonts that ruptures to release more merozoites. The rupturing of schizonts marks the start of malaria symptoms in the human host following the action of the Plasmodium parasite on the host’s cells to release cytokines.
This cycle is repeated for approximately 48 hours (for P. falciparum, P. vivax, and P. ovale) or 72 hours (for P. malariae) and parasitaemia continues depending on the infecting Plasmodium parasite. Sexual stage in the human host is usually characterized by the entering of some parasite merozoites into the erythrocytes. These merozoites later differentiate into male gametocytes (microgametocytes) and female gametocytes (macrogametocytes). This marks the beginning of the sexual phase in the human host, but this sexual stage of the Plasmodium development is not completed in humans but in the insect vector.
This occurs when the gametocytes (male and female) are picked up and ingested by female Anopheles mosquitoes during a blood meal; and this ingestion allows the insect vector to continue the transmission of the Plasmodium parasite to susceptible human hosts as it sucks and feed on their blood. It is noteworthy that not all merozoites released from RBCs can go on to infect other erythrocytes. The gametocytes (which are offshoots of merozoites) cannot infect the erythrocytes but rather infects only mosquitoes.
Sporogonic stage
The sporogonic phase occurs solely in the female Anopheles mosquito, and it is characterized by the multiplication of the Plasmodium parasite in the midgut (stomach) of the insect vector. This stage in the Plasmodium parasite life cycle begins immediately after the ingestion of the gametocytes by the female anopheles mosquito during a blood meal.
Inside the stomach of the mosquito, the male and female gametocytes undergo fertilization (usually meiosis and fusion of both cells) to form a zygote which later transforms into a motile and elongated form called ookinete. The ookinete enters the midgut wall of the mosquito’s stomach where they later develop into oocysts. The Plasmodium parasite usually takes about 10-35 days to develop within the insect vector (and this stage is called sporogony).
The oocysts mature and rupture to release plenty of sporozoites which migrate to the salivary glands of the female Anopheles mosquito where they wait to be transmitted to a susceptible human host during a blood meal. The life cycle of the Plasmodium parasite infection (malaria episode) becomes completed when the sporozoites successfully gain entry into the vascular system of a human host during a blood meal.
Genetics and molecular basis of erythrocyte invasion by Plasmodium
Plasmodium parasites are intracellular protozoa that have an intracellular existence within their affected host cells. Their ability to enter and modify the cellular and molecular mechanisms of their host cells is very advantageous to the Plasmodium parasites as this phenomenon gives them an edge to evade the host immune attack.
Merozoites, sporozoites and ookinetes are the three characteristic invasive forms of Plasmodium parasites. While merozoites occur at the blood stages of the Plasmodium parasite (erythrocytic and pre-erythrocytic stages inclusive), both the sporozoites and the ookinetes occur at the mosquito stage of the life cycle of Plasmodium species.
The intimate host-parasite relationship that exist between Plasmodium parasites and their human hosts allows them to acquire virtually all the nutrients, macromolecules and other biochemicals required for both anabolic and catabolic reactions from their human host.
Thus, Plasmodium parasites have a distinct metabolic pathway from that of their human hosts, and as such could be exploited in the design for novel antimalarial drugs to contain the disease (malaria) that they cause.
SIGNS AND SYMPTOMS OF MALARIA
Malaria is usually accompanied with some specific symptoms, and these include: fever that reaches 40oC or more, chills or cold, headache, nausea, fatigue, vomiting, diarrhea, occasional cough, abdominal pain, splenomegaly (enlargement of the spleen), hepatomegaly (enlargement of the liver), jaundice, chest pain, and myalgia (muscle pain).
Loss of appetite and bitterness of the mouth can also be experienced by some patients. Malaria symptoms are usually manifested in a human host following the rupturing of the red blood cells (erythrocytes) to release erythrocyte schizonts.
This action triggers immune response in the host which leads to the formation of cytokines and other immune system products that spark up an inflammatory action that usually results to the chills and fever experienced by the individual. It is noteworthy that the clinical manifestation of malaria is ushered in following the invasion of the erythrocyte by the Plasmodium parasite.
FACTORS THAT AFFECT THE RESULT OF MALARIA DIAGNOSIS
The clinical and laboratory diagnosis of malaria is challenging to health practitioners, physicians and scientists alike owing to some diagnostic complexity associated with the disease. Malaria is a febrile infection that usually presents clinically with signs and symptoms that are significantly related to other feverish infections (of bacterial or viral origin) common in most tropical regions where the disease is widespread.
To detect the parasite and administer therapy to the affected patient appropriately, it is vital that scientist take into consideration some of the prevailing factors that affect the reliability of malaria test results. Some of these factors which commonly promote the indiscriminate use of antimalarial drugs (a cause for emergence and spread of resistant Plasmodium strains) and hold back the specificity of identifying and correctly interpreting malaria parasitaemia diagnostic test results include:
- Administration of antimalarial drugs based only on clinical diagnosis.
- Presence of persisting viable or non-viable Plasmodium parasites in blood.
- Drug resistance of some Plasmodium species.
- Sequestration of Plasmodium parasites in host tissues.
- Movement of people from malaria-endemic regions to non-endemic areas and vice-versa.
- Endemicity of some Plasmodium species in some regions (P. falciparum in sub-Saharan Africa).
- Signs and symptoms, and diagnosis of malaria infection.
- Issues arising from reporting non-malarial febrile infections as malaria in endemic regions (Africa and Asia).
LABORATORY DIAGNOSIS OF MALARIA
Aside, laboratory diagnosis, clinical diagnosis of malaria is often practiced in some quarters to diagnose the disease but this practice is not versatile (though traditional amongst most physicians) due to its non-specificity and the possibility of missing out other febrile non-malarial infections in the process. The diagnosis of malaria in the hospital or laboratory is usually suspected when patients present with febrile conditions. Usually venous blood or capillary blood specimens are obtained from affected patients, and these are examined microscopically in the laboratory.
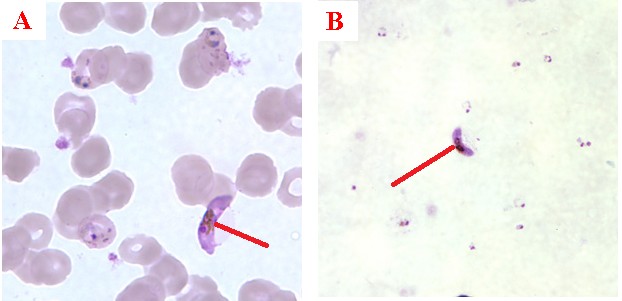
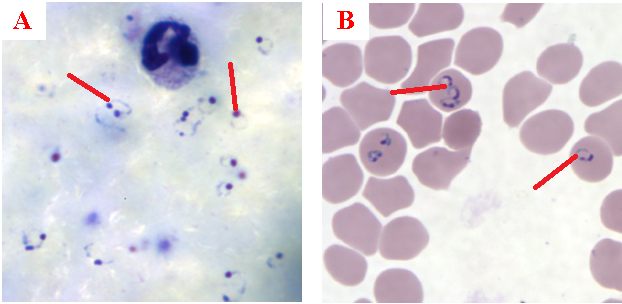
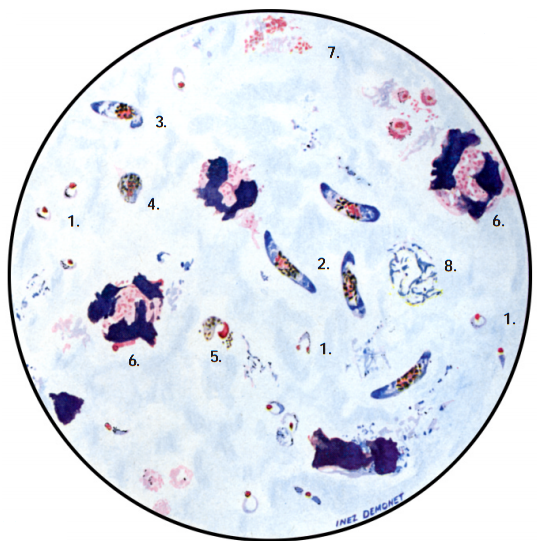
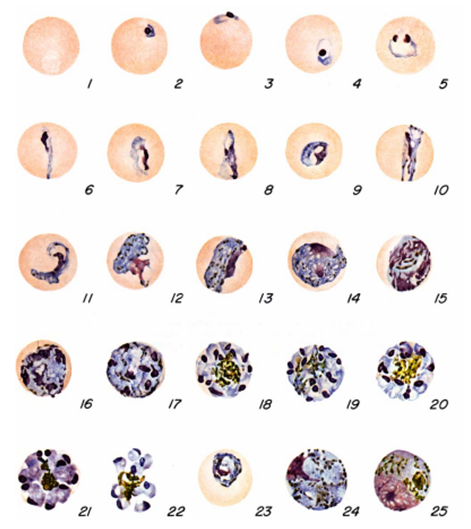
The diagnosis of malaria infection normally begins with the identification of Plasmodium parasites in the blood smears of patients. Thick and thin blood smears are usually made using the Giemsa staining technique. The ring forms of Plasmodium species as well as their trophozoites and gametocytes are often among the key features of the Plasmodium parasite looked for when performing microscopy on blood smears for detection of malaria parasitaemia.
The ring forms of Plasmodium species (particularly P. falciparum) are morphologically endowed withcytoplasm and one or two small chromatin dots; and P. falciparum gametocytes usually assumes a crescent or sausage shape when observed under the microscope (Figure 2). The trophozoites of P. falciparum assume an amoeboid shape (Figure 3), and they are rarely seen in peripheral blood smears.
While the thin blood smear helps to differentiate the different species of Plasmodium parasites, the thick blood film preparation helps to concentrate the parasite and help to detect mild cases of malaria parasitaemia. Serological techniques which involve the use of rapid diagnostic tests (containing monoclonal antibodies) to detect specific Plasmodium parasite antigens from blood specimens have also been introduced in malaria laboratory diagnosis. Most recently in the diagnosis of malaria parasitaemia is the use of molecular detection techniques including gene amplification methods, polymerase chain reaction (PCR) and nucleic acid probes to detect malaria parasites from blood specimens of patients. However, the Giemsa staining technique involving thick and thin blood films is still being used in most part of the developing nations as the primary diagnostic protocol for malaria parasitaemia. Abnormal liver function tests and an elevated lactate dehydrogenase tests coupled with low haemoglobin levels should also raise suspicion of malaria parasitaemia.
Overview of protocols used for the diagnosis of malaria infection
- Microscopy: Microscopy as earlier explained is this textbook is still the gold standard for the laboratory diagnosis of malaria infection in many parts of the world where the disease occurs. It usually involves the microscopical investigation of peripheral blood smear (PBS) – which can either be a thick smear (for parasite detection) or thin smear (for parasite-species identification). All the four species of Plasmodium i.e. P. falciparum (Figure 4), P. malariae(Figure 5), P. ovale (Figure 6) and P. vivax (Figure 7) that causes malaria in humans appear differently under the microscope when a thick blood smear is made and examined. And these morphological appearances of the Plasmodium species aids in their identification in the laboratory.
Microscopic technique though very relevant in the laboratory diagnosis of malaria is laborious, time-consuming and parasite-specie identification at low levels of parasitaemia is very challenging. This method requires well trained personnel for optimum result. Briefly, two types of blood specimens may be required for laboratory detection of malaria parasites using the microscopy technique: capillary blood obtained by fingerstick and venous blood obtained by venipuncture.
To obtain capillary blood, clean the middle or ring finger of the patient with 70 % alcohol; allow to dry. Puncture the ball of the finger and wipe away the first drop with a clean cotton wool. For infants, the heel is usually punctured to obtain venous blood. Touch the next drop of blood with the center of a clean glass labeled slide. Venous blood is usually obtained in an EDTA bottle after drawing blood from the patient using a sterile string.
For thin blood smear, place a drop of the blood at one end of a clean glass slide, and use another slide to push and spread the blood forward in a smooth and rapid fashion. For thick blood smear, place a drop of blood at the center of a clean glass slide, and use the edge of another slide to spread the blood in a circular fashion while making sure not to make it too thick. Allow the thin and thick blood smears to dry properly before staining. Thin blood smear is fixed with methanol prior to staining but thick blood smear is not fixed before staining.
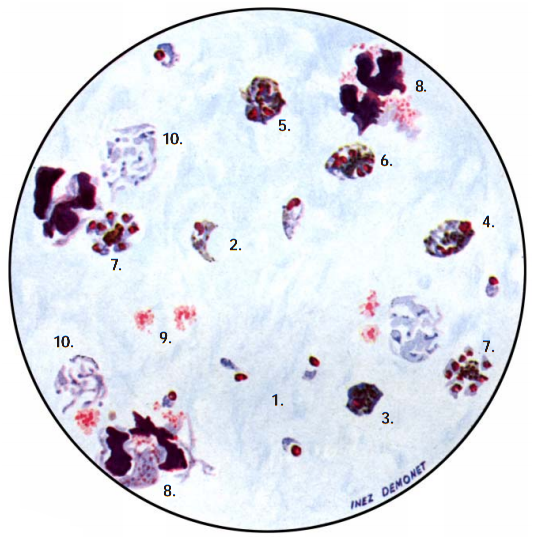

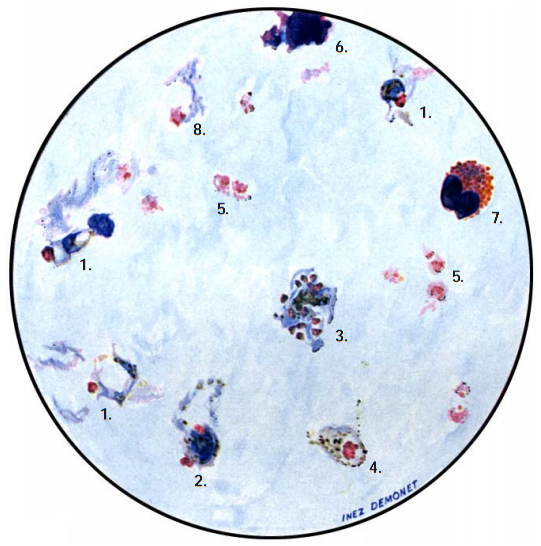
Note: Allow the fixed thin film to dry before staining. Giemsa stain is usually used for staining blood smears required for detection of Plasmodium parasites, and stained smears are viewed under oil immersion objective lens. In thin smear preparations, the morphological appearances of the components of the Plasmodium species i.e. P. falciparum (Figure 8), P. malariae(Figure 9), P. ovale (Figure 10) and P. vivax (Figure 11) also differ slightly from their appearances in a thick smear preparation.
- Rapid diagnostic tests (RDTs): RDTs are immunochromatographic techniques that employ specialized malaria detection test kits for the laboratory diagnosis of malaria infection. They are easy to use and very reliable in the prompt detection of malaria parasites from patient’s specimens.
Unlike the microscopic technique, RDTs does not require any specialized training as they can be used based on the manufacturers instruction. RDTs are cost-effective and they were developed to take care of some of the lapses associated with microscopy such as time wastage and inability to detect parasites at low levels of parasitaemia. Some of the commercially available RDTs include ParaSight F, ICT Malaria Pf, and OptiMAL amongst others.
- Quantitative Buffy Coat (QBC): The QBC technique is a centrifugation method used for the laboratory diagnosis of malaria infection. In this method, blood specimens are centrifuged in specialized tubes (e.g. haematocrit tubes) containing anticoagulant such as EDTA and acridine orange. After centrifugation, the tubes are examined under a fluorescent microscope to detect Plasmodium nuclei or DNA and other parasite components.

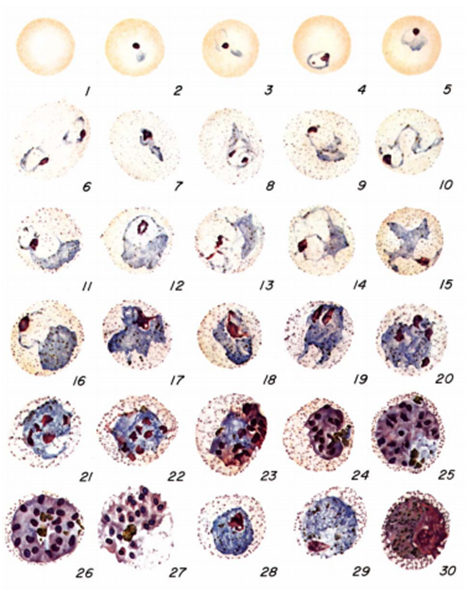
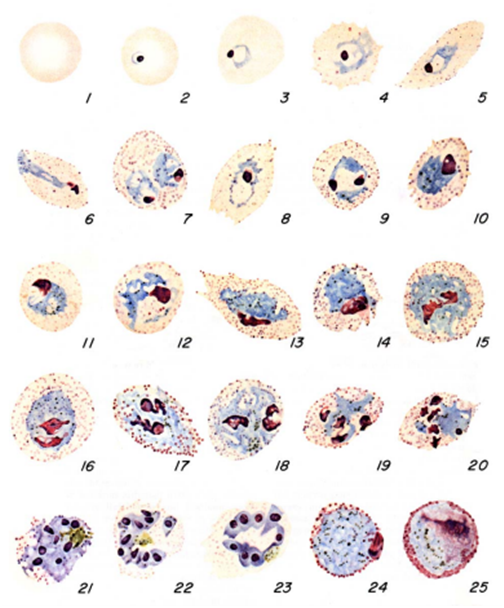
The acridine orange stains the DNA of the parasite during centrifugation, and it appears as a fluorescing bright-green matter under the fluorescent microscope. The cytoplasm appears as a yellow-orange matter. QBC technique is not versatile in the laboratory detection of malaria infection due to its high cost and inability or poor performance to detect parasite species and numbers.
- Serology: Serological tests have also been employed in the laboratory diagnosis of malaria infection. They are mainly based on the detection of specific antibodies produced against the malaria parasite in the blood (serum) of infected individuals. Immunoflourescence antibody testing (IFA) which employs antigen-antibody reactions is one of the versatile methods used in most serological tests.
- Polymerase Chain Reaction (PCR): PCR is the most sensitive and specific technique for the diagnosis of malaria infection as it can differentiate between mixed infection and actual malaria infection. Unlike other techniques, PCR can detect malarial parasites at very low levels of parasitaemia at the molecular level.
However, its usage for the routine diagnosis of malaria in most malaria-endemic regions has been limited by unavailability of specialized trained personnel, high cost of purchase and maintenance, and its mode of operation is more complex than the microscopic technique and other methods used for the laboratory diagnosis of malaria infection.
- Other tests: Most traditional methods of diagnosing malaria though versatile and inexpensive to perform are usually problematic and challenging. Some of these methods (for example clinical diagnosis) are unreliable while others require a well trained health personnel and equipments which may be lacking in some local communities where the incidence of malaria infection is high.
Prompt detection and diagnosis of malaria infection in places where Plasmodium parasite transmission is widespread is vital to the reduction of the morbidity and mortality due to the disease. The challenges associated with some of the traditional methods of diagnosis and detecting malaria parasites in the laboratory and clinical settings have stepped up the need for newer and better detection protocols with high specificity and sensitivity for parasite detection.
These newer techniques used for the diagnosis of malaria infection detect malaria parasites from patient’s specimens at the molecular level; and they include: real-time PCR technique, reverse transcription PCR, nested PCR, and DNA microarrays. Though very reliable, specific and sensitive in detecting malaria parasites from patient’s specimen; some of these techniques are not versatile and available for the routine diagnosis of malaria infections in most regions of developing countries where the disease is prevalent.
They require specialized trained personnel and high cost of maintenance. Thus, some of these techniques are only practiced or used in reference laboratories for the detection of malaria parasites. ELISA, parasite culture techniques, enzyme immunoassay, flow cytometry, LAMP technique and mass spectrophotometry are other non-molecular techniques which are currently been employed for the prompt detection and diagnosis of malaria infection.
TREATMENT OF MALARIA
The bite of an infected female Anopheles mosquito is required for the transmission of the Plasmodium parasite that causes malaria, thus preventing the bite of this mosquito will help to prevent the acquisition and transmission of the disease. There have been several reported cases of resistance of malaria parasites to some available antimalarial drugs even though artemisinin-based combination therapy (ACT) is still the drug of choice for malaria treatment.
However, the treatment of malaria infection usually involves a series of combination therapy that incorporates one antimalarial agent and another as a way of fighting the resistance of the parasite while ensuring a better prognosis in the patient taking the medication. There are usually some variations in the treatment of malaria infection depending on the region where the infection occurred and the prevalent Plasmodium parasite in that geographical area.
Thus, there abound certain national guidelines and measures adopted for the effective treatment of the disease depending on the endemicity of the disease in the region. Malaria chemotherapy usually involves the use of Artemisinin-related compounds (artesunate, artemether, and artemisinin), Quinolone-related compounds (mefloquine, chloroquine, primaquine, and quinine), Anti-folates (trimethoprim, pyrimethamine, and proguanil), and certain Antibacterial agents such as macrolides, tetracyclines, and sulphonamides which are used in combination with antimalarial drugs when malaria therapy is anticipated.
Chloroquine (which was among the first antimalarial agent), which can be administered either orally or parenterally still remains the preferred drug option for all forms of malaria infection except for certain stages of P. falciparum malaria infection. Some antimalarial drugs are contraindicated in pregnancy and even in some disease condition (cardiac disorders and disorders of the renal system), thus malaria treatment in pregnant women should be undertaken with caution to prevent fatality in the foetus or mother.
Antimalarial agents that functions as folate-antagonists (proguanil) helps to block the synthesis of folate (folic acid) in Plasmodium parasites, which is an important process required by the parasite to thrive. Plasmodium parasites are incapable of utilizing pyrimidines from their human host, thus an obstruction of their innate folate synthesis machinery will automatically result to cell death due to a reduction in pyrimidines and other nitrogenous bases required for synthesis of the organism’s nucleic acid.
Artemisinin-related agents are a class of drugs which were originally sourced from a Chinese herbal plant called Artemisia annua(Qinghaosu); and these drugs have been found to be effective to the different forms of malaria infection including multidrug-resistant P. falciparum malaria infection. Quinolone-related compounds (quinine) used for the treatment of malaria infection are alkaloids sourced from the bark of Cinchona tree, and they have been found to be effective against P. falciparum infection.
Sulphonamides and other antibacterial agents administered in combination with other antimalarial drugs are used as prophylactics and also for the treatment of P. falciparum malaria infection. These agents usually act as folate-antagonist, as they inhibit the synthesis of folic acid by the Plasmodium parasite.
PREVENTIVE AND CONTROL MEASURES FOR MALARIA
Malaria should be controlled and prevented in human populations (especially in the subtropics and tropical regions where its dominance is evident) due to the economic and medical consequences and burden of the disease. It is a life-threatening parasitic infection that requires complete cure.
Controlling malaria infection will help to reduce the morbidity and mortality due to the disease; improve mans activities; reduce and channel resources spent on malaria treatment for other useful ventures, and most of all, it will ensure a better life and future for the present and future generations of individuals in malaria endemic regions. A reduction in malaria morbidity and mortality especially amongst high-risk groups for example pregnant women, breastfeeding mothers, and children through adequate and sustainable containment of the transmission of the disease through the bite of female Anopheles mosquito is critical in malaria endemic regions.
However, the world through its many malaria control and eradication programs such as the Roll Back Malaria (RBM) Programme and the rest are looking at a total eradication of the disease in those parts of the world where malaria still accounts for a high rate of morbidity and mortality amongst high-risk groups.
The use of artemisinin-based combination therapy (ACT) in addition to the protection of women with intermittent preventive treatment during pregnancy (IPTp) has helped to reduce the health and economic menace of the disease in African continent. Also, the use of long-lasting insecticide-treated mosquito nets (LLITNs) and indoor residual spraying (IRS) using potent insecticides have also been recommended as preventive and control measures for the disease.
Malaria is a widespread mosquito-borne disease caused by Plasmodium parasite which is transmitted through the bite of an infected mosquito. The disease is among the leading cause of death in the tropic and subtropical parts of the world; and its prevention is dependent upon strict personal hygiene, effective chemotherapy and a holistic public health measures towards the disease.
Malaria disease which already has enormous negative impact on humanity should be prevented and controlled as much as possible in order to ensure better health for people in malaria endemic countries. The total prevention and control of malaria in the subtropics and tropical counties of the world is still a mirage. Eradication of both the Plasmodium parasite and its insect vector in these areas has been very much slower.
The control and prevention of malaria depends hugely on the total elimination of possible breeding sites of the insect vector (female Anopheles mosquito) and avoidance of long biting contact between human hosts and the insect vector. The resistance of the Plasmodium parasite to readily available antimalarial drugs coupled with the prevailing resistance of the insect vector to insecticides have further compounded the established control measures for the disease in affected parts of the world.
Malaria is a vector-borne disease that is transmitted from person to person through the female Anopheles mosquito; thus, protecting people from the insect vector (female Anopheles mosquito) and reducing the number of insect vectors in human populations/environment will go a long way in helping to stop the spread of the disease. There is still no vaccine against the disease; nevertheless, frantic efforts to develop an effective malaria vaccine are still in progress and very promising.
Also, existing malaria control and preventive efforts (including the use of LLITNs, IRS, and IPTp) in malaria endemic regions are beginning to show promising results in some countries, and this has helped to lower the morbidity and mortality associated with the disease in these areas. There are significant variations amongst countries that use LLITNs as a malaria control measure. The reason that may be attributed to the poor distribution or usage of LLITNs in malaria endemic countries in Africa may be connected to paucity of resources to make these nets available for use.
In some cases, some people may have it but still do not appreciate the importance of sleeping under the nets, and thus will resort to not using them at all. Whatever maybe the reason for the non-usage of LLITNs by children in some malaria endemic regions of Africa as depicted in the figure below, advocacy and education is a vital tool to convincing these people to the benefit of using these nets as one of the ways to be malaria free. Resources should be channeled towards research and development (R&D) in malaria endemic countries in order to develop novel ways of fighting the disease so that the world’s target of completely eradicating malaria can be achieved.
OTHER PREVENTIVE AND CONTROL MEASURES FOR MALARIA
- Ensuring regular and effective insecticide spraying around homes and other mosquito breeding sites in order to destroy the insect vectors.
- Constant drainage of swamps and possible destruction or elimination of other breeding sites of the mosquito’s habitats.
- Avoiding mosquito bites by wearing long clothes to cover the whole body when in endemic areas, use of long lasting insecticide treated mosquito nets and use of mosquito nettings on doors and windows of houses to prevent the invasion of the insect vector.
- Drainage of breeding sites such as gutters to remove surface waters and filling of ponds and pot-holes with sands to prevent breeding of the insect vector.
- Use of effective anti-malarial drugs to treat patients during an infection in order to break the life cycle of the Plasmodium parasite.
- Proper funding of preventive programs (such as the Roll Back Malaria Initiative of the WHO) in endemic areas couple with the stepping up of research developments of malaria vaccine and a lasting cure or treatment to the malady is also vital to the termination of the disease.
- Local research in malaria endemic areas should be strengthened as a way of finding a lasting solution to the malaria endemicity.
- People travelling to malaria endemic areas should avoid mosquito bites as much as possible by sleeping under long lasting insecticide treated nets and covering the body properly when outdoor.
References
Aschengrau A and Seage G.R (2013). Essentials of Epidemiology in Public Health. Third edition. Jones and Bartleh Learning,
Beers M.H., Porter R.S., Jones T.V., Kaplan J.L and Berkwits M (2006). The Merck Manual of Diagnosis and Therapy. Eighteenth edition. Merck & Co., Inc, USA.
Chiodini P.L., Moody A.H., Manser D.W (2001). Atlas of medical helminthology and protozoology. 4th ed. Edinburgh: Churchill Livingstone.
Dictionary of Microbiology and Molecular Biology, 3rd Edition. Paul Singleton and Diana Sainsbury. 2006, John Wiley & Sons Ltd. Canada.
Ghosh S (2013). Paniker’s Textbook of Medical Parasitology. Seventh edition. Jaypee Brothers Medical Publishers,
Gillespie S.H and Pearson R.D (2001). Principles and Practice of Clinical Parasitology. John Wiley and Sons Ltd. West Sussex, England.
Gordis L (2013). Epidemiology. Fifth edition. Saunders Publishers, USA.
John D and Petri W.A Jr (2013). Markell and Voge’s Medical Parasitology. Ninth edition.
Kumar V, Abbas A.K, Fausto N and Aster A (2009). Robbins and Cotran Pathologic Basis of Disease. 8th edition. W.B. Saunders Co, USA.
Lee JW (2005). Public health is a social issue. Lancet. 365:1005-6.
Leventhal R and Cheadle R.F (2013). Medical Parasitology. Fifth edition. F.A. Davis Publishers,
Lucas A.O and Gilles H.M (2003). Short Textbook of Public Health Medicine for the tropics. Fourth edition. Hodder Arnold Publication, UK.
MacMahon B., Trichopoulos D (1996). Epidemiology Principles and Methods. 2nd ed. Boston, MA: Little, Brown and Company. USA.
Mandell G.L., Bennett J.E and Dolin R (2000). Principles and practice of infectious diseases, 5th edition. New York: Churchill Livingstone.
Molyneux, D.H., D.R. Hopkins, and N. Zagaria (2004) Disease eradication, elimination and control: the need for accurate and consistent usage. Trends Parasitol, 20(8):347-51.
Nelson K.E and Williams C (2013). Infectious Disease Epidemiology: Theory and Practice. Third edition. Jones and Bartleh Learning
Roberts L, Janovy J (Jr) and Nadler S (2012). Foundations of Parasitology. Ninth edition. McGraw-Hill Publishers, USA.
Schneider M.J (2011). Introduction to Public Health. Third edition. Jones and Bartlett Publishers, Sudbury, Massachusetts, USA.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.