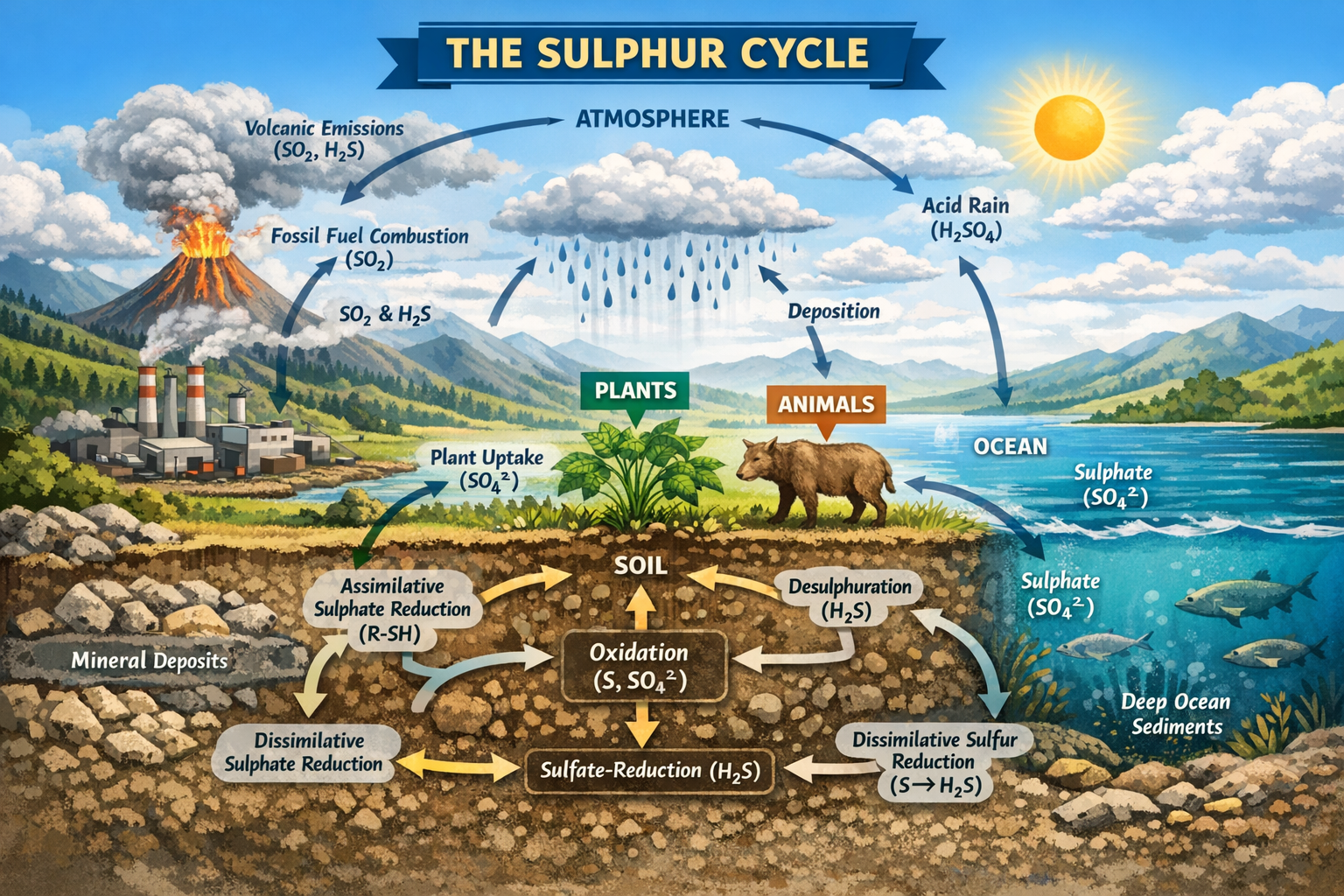
The sulphur cycle is a fundamental biogeochemical cycle that regulates the movement, transformation, and availability of sulphur across the Earth’s major environmental compartments: the biosphere, lithosphere, atmosphere, and hydrosphere. Unlike purely geological cycles that operate over millions of years, the sulphur cycle functions across a wide range of temporal and spatial scales, from rapid microbial transformations in soils and sediments to long-term storage in rocks and oceanic deposits. Sulphur is indispensable to life, industrial development, and ecosystem stability, yet it is also associated with some of the most significant environmental challenges, including air pollution, acid rain, and soil degradation. Understanding and actively managing the sulphur cycle is therefore essential for sustaining agricultural productivity, protecting ecosystems, and mitigating anthropogenic environmental impacts.
Sulphur plays a critical biochemical role in living organisms. It is a structural component of the sulphur-containing amino acids cysteine and methionine, which are essential for protein synthesis. It is also present in vitamins (such as biotin and thiamine), coenzymes, hormones, and various metabolic intermediates. In plants, sulphur is necessary for chlorophyll formation, enzyme activation, and resistance to stress. In animals and humans, sulphur supports protein structure, detoxification pathways, and normal physiological functioning. Because animals obtain sulphur primarily through plant-based diets, the availability of sulphur in soils directly affects food quality and nutritional health.
Beyond its biological importance, sulphur and its compounds are central to industrial processes. Sulphuric acid is one of the most widely produced industrial chemicals globally, serving as a key input in fertilizer manufacture, petroleum refining, mineral processing, and chemical synthesis. Sulphur dioxide is used as a bleaching agent, preservative, and disinfectant. However, the same compounds that provide economic value can become serious environmental pollutants when released in excess into the atmosphere. Consequently, the sulphur cycle represents a complex intersection of natural processes, biological requirements, and human activities.
Forms and Distribution of Sulphur in the Earth System
Sulphur exists in multiple oxidation states, ranging from highly reduced forms such as hydrogen sulphide (H₂S, –2 oxidation state) to highly oxidized forms such as sulphate (SO₄²⁻, +6 oxidation state). This chemical versatility underpins the diversity of sulphur transformations in nature. In the lithosphere, sulphur is largely stored in sedimentary rocks, sulphide minerals (e.g., pyrite, FeS₂), sulphate minerals (e.g., gypsum, CaSO₄·2H₂O), and deep-sea sediments. These geological reservoirs contain the majority of the Earth’s sulphur and release it slowly through weathering, erosion, and volcanic activity.
In the hydrosphere, sulphur is predominantly present as dissolved sulphate in oceans, rivers, and groundwater. Seawater represents one of the largest active sulphur reservoirs, with sulphate concentrations that support marine primary productivity and microbial sulphur cycling. In the atmosphere, sulphur occurs mainly as sulphur dioxide (SO₂), hydrogen sulphide (H₂S), sulphur trioxide (SO₃), and sulphate aerosols. Although the atmospheric pool is relatively small compared to geological reservoirs, it plays a disproportionately large role in global sulphur redistribution due to rapid transport and deposition processes.
The biosphere contains sulphur primarily in organic forms, incorporated into proteins, enzymes, and other biomolecules. Living organisms continuously exchange sulphur with their environment through uptake, assimilation, decomposition, and excretion. Soil acts as a critical interface where geological, biological, and atmospheric sulphur pools interact, making terrestrial ecosystems particularly sensitive to changes in sulphur inputs and transformations.
Biological Importance of Sulphur
Sulphur is essential for the structure and function of proteins because disulphide bonds between cysteine residues contribute to protein folding and stability. Many enzymes require sulphur-containing cofactors to facilitate redox reactions, electron transport, and metabolic regulation. In plants, sulphur deficiency leads to reduced growth, chlorosis, delayed maturity, and poor yield quality. Sulphur is especially important in oilseed crops and legumes, where it supports nitrogen fixation and protein synthesis.
Animals depend on sulphur indirectly through plant consumption. Sulphur-containing amino acids are essential in animal nutrition, influencing growth, immune function, and tissue repair. In humans, sulphur is involved in detoxification processes in the liver, antioxidant defense (via glutathione), and structural integrity of connective tissues. Thus, disruptions in the sulphur cycle that reduce sulphur availability in soils can cascade through food webs and affect ecosystem and human health.
Major Processes in the Sulphur Cycle
The sulphur cycle is driven by a series of interrelated physical, chemical, and biological processes that convert sulphur between organic and inorganic forms and between different oxidation states. Microorganisms play a central role in mediating most of these transformations, particularly in soils, sediments, and aquatic environments.
Mineralization of Organic Sulphur
Mineralization is the process by which organic sulphur compounds in plant and animal residues are decomposed by soil microorganisms, releasing inorganic sulphur forms such as hydrogen sulphide (H₂S) or sulphate. Proteins containing cysteine and methionine are broken down into amino acids, which are further degraded to release sulphur. This process is critical for recycling sulphur and maintaining its availability for plant uptake.
Assimilative Sulphate Reduction
Assimilative sulphate reduction occurs in plants, fungi, and many prokaryotes. In this pathway, sulphate (SO₄²⁻) absorbed from soil or water is enzymatically reduced and incorporated into organic sulphur compounds, particularly amino acids and proteins. This process does not release sulphur back into the environment immediately but stores it in biomass until decomposition occurs.
Desulphuration
Desulphuration involves the removal of sulphur from organic molecules, resulting in the release of hydrogen sulphide gas. This process occurs during microbial decomposition under both aerobic and anaerobic conditions and contributes to the pool of reduced sulphur compounds in soils and sediments.
Oxidation of Reduced Sulphur Compounds
Hydrogen sulphide and elemental sulphur can be oxidized to sulphate by sulphur-oxidizing bacteria, particularly under aerobic conditions. These microorganisms use reduced sulphur compounds as energy sources, linking sulphur transformations to microbial metabolism and ecosystem energy flow. Oxidation processes are especially important in well-aerated soils and surface waters.
Dissimilative Sulphur and Sulphate Reduction
Under anaerobic conditions, certain microorganisms use sulphate or elemental sulphur as terminal electron acceptors in respiration, producing hydrogen sulphide. This dissimilative sulphate reduction is common in waterlogged soils, sediments, and wetlands. The hydrogen sulphide produced can react with metals to form insoluble sulphides or be re-oxidized when conditions become aerobic, thereby continuing the cycle.
The Role of Soil and Microorganisms
Soil is the primary terrestrial arena for sulphur cycling. Most soil sulphur exists in organic forms, bound within soil organic matter. Microbial communities regulate the balance between immobilization (incorporation into microbial biomass) and mineralization (release into inorganic forms). Environmental factors such as soil moisture, temperature, pH, and oxygen availability strongly influence these processes.
Sulphur-reducing bacteria and sulphur-oxidizing bacteria form complementary functional groups that drive sulphur transformations. Their activities link the sulphur cycle to other biogeochemical cycles, particularly the carbon and nitrogen cycles. For example, sulphate reduction is often coupled with the oxidation of organic carbon, while sulphur oxidation can influence soil acidity and nutrient availability.
Atmospheric Sulphur and Deposition
Sulphur enters the atmosphere through both natural and anthropogenic sources. Natural emissions include volcanic eruptions, sea spray, and the decomposition of organic matter, which release sulphur dioxide and hydrogen sulphide. Human activities, particularly the combustion of fossil fuels such as coal, oil, and natural gas, have dramatically increased atmospheric sulphur emissions over the past two centuries.
In the atmosphere, sulphur dioxide undergoes oxidation to form sulphur trioxide and sulphate aerosols. These compounds can react with water vapor to produce sulphuric acid, which returns to the Earth’s surface through wet deposition (acid rain) or dry deposition. Atmospheric transport allows sulphur emissions to affect regions far from their source, making sulphur pollution a transboundary environmental issue.
Environmental Impacts of Sulphur Imbalance
Excessive sulphur deposition has significant ecological consequences. Acid rain lowers soil pH, leaches essential nutrients such as calcium and magnesium, and mobilizes toxic metals like aluminum. These changes impair plant growth, reduce microbial activity, and disrupt soil structure. Aquatic ecosystems are particularly vulnerable, as acidification can lead to fish mortality, reduced biodiversity, and altered food webs.
Conversely, sulphur deficiency has emerged as an agricultural concern in some regions due to reduced atmospheric deposition following air pollution control measures. Modern high-yield crop varieties and intensive farming systems often require supplemental sulphur fertilization to maintain productivity and crop quality. This highlights the need for balanced sulphur management rather than simply minimizing emissions.
Human Activities and the Modern Sulphur Cycle
Industrialization has profoundly altered the natural sulphur cycle. Mining, fossil fuel combustion, industrial processing, and large-scale agriculture have increased sulphur mobilization and redistribution. While regulatory measures have successfully reduced sulphur dioxide emissions in many countries, legacy effects persist in soils and ecosystems previously exposed to high sulphur loads.
Sulphuric acid production underpins fertilizer manufacturing, linking the sulphur cycle directly to global food systems. At the same time, inappropriate management of industrial sulphur emissions can exacerbate environmental degradation. Sustainable development therefore requires integrated strategies that balance economic benefits with ecological protection.
Managing the Sulphur Cycle for Sustainability
Effective management of the sulphur cycle involves coordinated actions across agriculture, industry, and environmental policy. In agricultural systems, soil testing and targeted sulphur fertilization can prevent both deficiency and excess. Incorporating organic matter, promoting microbial diversity, and improving soil aeration enhance natural sulphur cycling processes.
In industrial and energy sectors, emission controls, cleaner fuel technologies, and sulphur recovery systems reduce atmospheric pollution. Environmental monitoring and international cooperation are essential to address transboundary sulphur deposition. At the ecosystem level, restoration of degraded soils and wetlands can re-establish balanced sulphur transformations and improve resilience.
The sulphur cycle is a dynamic and interconnected system that underpins biological productivity, industrial development, and environmental quality. Sulphur’s essential role in proteins, enzymes, and metabolic processes makes it indispensable to life, while its chemical versatility drives complex transformations across the Earth system. Human activities have significantly altered sulphur flows, creating both challenges and opportunities for sustainable management.
A comprehensive understanding of the sulphur cycle highlights the importance of microbial processes, soil health, atmospheric dynamics, and responsible industrial practices. Managing the sulphur cycle proactively is not merely an academic concern; it is a practical necessity for ensuring food security, protecting ecosystems, and supporting sustainable development in an increasingly industrialized world.
References
Abrahams P.W (2006). Soil, geography and human disease: a critical review of the importance of medical cartography. Progress in Physical Geography, 30:490-512.
Ahring B.K, Angelidaki I and Johansen K (1992). Anaerobic treatment of manure together with industrial waste. Water Sci. Technol, 30, 241–249.
Andersson L and Rydberg L (1988). Trends in nutrient and oxygen conditions within the Kattegat: effects on local nutrient supply. Estuar. Coast. Shelf Sci, 26:559–579.
Ballantyne A.P, Alden C.B, Miller J.B, Tans P.P and White J.W.C (2012). Increase in observed net carbon dioxide uptake by land and oceans during the past 50 years. Nature, 488: 70-72.
Baumgardner D.J (2012). Soil-related bacterial and fungal infections. J Am Board Fam Med, 25:734-744.
Paul E.A (2007). Soil Microbiology, ecology and biochemistry. 3rd edition. Oxford: Elsevier Publications, New York.
Pelczar M.J Jr, Chan E.C.S, Krieg N.R (1993). Microbiology: Concepts and Applications. McGraw-Hill, USA.
Pelczar M.J., Chan E.C.S. and Krieg N.R. (2003). Microbiology of Soil. Microbiology, 5th Edition. Tata McGraw-Hill Publishing Company Limited, New Delhi, India.
Pepper I.L and Gerba C.P (2005). Environmental Microbiology: A Laboratory Manual. Second Edition. Elsevier Academic Press, New York, USA.
Roberto P. Anitori (2012). Extremophiles: Microbiology and Biotechnology. First edition. Caister Academic Press, Norfolk, England.
Salyers A.A and Whitt D.D (2001). Microbiology: diversity, disease, and the environment. Fitzgerald Science Press Inc. Maryland, USA.
Sawyer C.N, McCarty P.L and Parkin G.F (2003). Chemistry for Environmental Engineering and Science (5th ed.). McGraw-Hill Publishers, New York, USA.
Ulrich A and Becker R (2006). Soil parent material is a key determinant of the bacterial community structure in arable soils. FEMS Microbiol Ecol, 56(3):430–443.
Discover more from Microbiology Class
Subscribe to get the latest posts sent to your email.